
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A INFORMATION
Proxy Statement Pursuant to Section 14(a) of
the Securities Exchange Act of 1934 (Amendment No. )
Filed by the Registrant x
Filed by a Party other than the Registrant ¨
Check the appropriate box:
| ¨ | Preliminary Proxy Statement | ¨ | Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2) | |||
| x | Definitive Proxy Statement | |||||
| ¨ | Definitive Additional Materials | |||||
| ¨ | Soliciting Material Pursuant to §240.14a-12 |
Bristol-Myers Squibb Company
(Name of Registrant as Specified In Its Charter)
(Name of Person(s) Filing Proxy Statement, if other than the Registrant)
Payment of Filing Fee (Check the appropriate box):
| x | No fee required | |||
| ¨ | Fee computed on table below per Exchange Act Rules 14a-6(i)(4) and 0-11 | |||
| (1) | Title of each class of securities to which transaction applies: | |||
| ||||
| (2) | Aggregate number of securities to which transaction applies: | |||
| ||||
| (3) | Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): | |||
| ||||
| (4) | Proposed maximum aggregate value of transaction: | |||
| ||||
| (5) | Total fee paid: | |||
| ||||
| ¨ | Fee paid previously with preliminary materials. | |||
| ¨ | Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. | |||
| (1) | Amount Previously Paid: | |||
| ||||
| (2) | Form, Schedule or Registration Statement No.: | |||
| ||||
| (3) | Filing Party: | |||
| ||||
| (4) | Date Filed: | |||
| ||||

March 22, 201021, 2011
NOTICEOF
MEETINGAND
PROXY STATEMENT
TUESDAY, MAY
AT 10:00 A.M.
BRISTOL-MYERS
SQUIBB COMPANY
777 SCUDDERS MILL RD.
PLAINSBORO
NEW JERSEY | DEAR FELLOW STOCKHOLDER:
You are cordially invited to attend the Annual Meeting of Stockholders of Bristol-Myers Squibb Company at
These materials include the Notice of Annual Meeting and the Proxy Statement. The Proxy Statement describes the business to be transacted at the meeting and provides other information about the company that you should know when you vote your shares.
The principal business of the Annual Meeting will • the election of • the ratification of the appointment of an independent registered public accounting • an advisory vote on the • an advisory vote on the frequency of the advisory vote on the compensation of our named executive officers; and • the consideration of three stockholder proposals. We will also review the status of the company’s business at the meeting.
Last year, over
Please follow the instructions in the Proxy Statement on how to attend the Annual Meeting. Admission to the Annual Meeting will be by ticket only.Please bring photo
We have provided space on the proxy card for comments from our registered stockholders. We urge you to use it to let us know your feelings about | |||||||
JAMES M. CORNELIUS Chairman of the Board |
LAMBERTO ANDREOTTI Chief Executive Officer | |||||||

345 Park Avenue
New York, New York 10154-0037
NOTICE OF ANNUAL MEETING
OF STOCKHOLDERS
Notice is hereby given that the 20102011 Annual Meeting of Stockholders will be held at Bristol-Myers Squibb Company, 777 Scudders Mill Road, Plainsboro, New Jersey, on Tuesday, May 4, 2010,3, 2011, at 10:00 a.m. for the following purposes as set forth in the accompanying Proxy Statement:
to elect to the Board of Directors the eleventwelve persons nominated by the Board, each for a term of one year;
to ratify the appointment of Deloitte & Touche LLP as the company’s independent registered public accounting firm for 2010;2011;
to approve three amendments conduct an advisory vote on the compensation of our named executive officers;
to conduct an advisory vote on the frequency of the advisory vote on the compensation of our Amended and Restated Certificate of Incorporation;named executive officers;
to consider three stockholder proposals, if presented at the meeting; and
to transact such other business as may properly come before the meeting or any adjournments thereof.
Holders of record of our common and preferred stock at the close of business on March 11, 2010,10, 2011, will be entitled to vote at the meeting.
By Order of the Board of Directors

SANDRA LEUNG
General Counsel and Secretary
Dated: March 22, 201021, 2011
YOUR VOTE IS IMPORTANT
Regardless of the number of shares you own, your vote is important. If you do not attend the Annual Meeting to vote in person, your vote will not be counted unless a proxy representing your shares is presented at the meeting. To ensure that your shares will be voted at the meeting, please vote in one of these ways:
| (1) | GO TO WWW.PROXYVOTE.COM and vote via the Internet; |
| (2) | CALL THE TOLL-FREE TELEPHONE NUMBER (800) 690-6903 (this call is toll-free in the United States); or |
| (3) | MARK, SIGN, DATE AND PROMPTLY RETURN the enclosed proxy card in the postage-paid envelope. |
If you do attend the Annual Meeting, you may revoke your proxy and vote by ballot.

PROXY STATEMENT
| Page | ||||
| 1 | ||||
| 5 | ||||
| 7 | ||||
| 7 | ||||
| 8 | ||||
| 8 | ||||
| 8 | ||||
| 8 | ||||
| 11 | ||||
| 11 | ||||
| 12 | ||||
| 12 | ||||
| 12 | ||||
| 13 | ||||
| 19 | ||||
| 19 | ||||
| 20 | ||||
| 21 | ||||
| 21 | ||||
| 26 | ||||
| 26 | ||||
| 46 | ||||
| 47 | ||||
| 48 | ||||
| 49 | ||||
| 51 | ||||
| 53 | ||||
| 54 | ||||
| 56 | ||||
| 57 | ||||
ITEMS TO BE VOTED UPON | ||||
| 13 | ||||
Item 2 — Ratification of the Appointment of Independent Registered Public Accounting Firm | 62 | |||
| 62 | ||||
Pre-Approval Policy for Services Provided by our Independent Registered Public Accounting Firm | 63 | |||
| 63 | ||||
| 65 | ||||
| Inside Back Cover |
GENERAL INFORMATION ABOUT THE ANNUAL MEETING AND VOTING
Why am I receiving these materials?
This Proxy Statement is being delivered to all stockholders of record as of the close of business on March 11, 201010, 2011 in connection with the solicitation of proxies on behalf of the Board of Directors for use at the Annual Meeting of Stockholders on May 4, 2010.3, 2011. Although the Annual Report and Proxy Statement are being delivered together, the Annual Report should not be deemed to be part of the Proxy Statement.
What is “Notice and Access” and how does it affect me?
The SEC has adopted a “Notice and Access” model which permits us to provide proxy materials to our stockholders electronically by posting the proxy materials on a publicly accessible website. Delivering proxy materials electronically will save us money by reducing printing and mailing costs. Accordingly, we have decided thatsent to all stockholders who hold fewer than 1,000 shares of our common and preferred stock will receiveand have not requested paper copies of our proxy materials a “Notice of Internet Availability of Proxy Materials.” This Notice will provideprovides instructions on how to access our proxy materials online and, if you prefer receiving a paper copy of the proxy materials, how you can request one. Employees and pension plan participants who have given consent to receive materials electronically will receivereceived a link to access our proxy materials by email. If you hold 1,000 or more shares and have not previously elected to receive materials electronically or if you are a pension plan participant who did not provide consent to receive materials electronically, you will bewere mailed a paper copy of the proxy materials. We encourage all of our stockholders who currently receive paper copies of the proxy materials to elect to view future proxy materials electronically if they have Internet access. You can do so by following the instructions when you vote your shares online or, if you are a beneficial holder, by asking your bank, broker or other holder of record how to receive proxy materials electronically.
What is “householding” and how does it work?
“Householding” is a procedure we adopted whereby stockholders of record who have the same last name and address and who receive the proxy materials by mail will receive only one copy of the proxy materials unless we have received contrary instructions from one or more of the stockholders. This procedure reduces printing and mailing costs. If you wish to receive a separate copy of the proxy materials, now or in the future, at the same address, or if you are currently receiving multiple copies of the proxy materials at the same address and wish to receive a single copy, you may contact us by writing to Stockholder Services, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, or by calling us at (212) 546-3309.
If you are a beneficial owner (your shares are held in the name of a bank, broker or other holder of record), the bank, broker or other holder of record may deliver only one copy of the Proxy Statement and Annual Report, or Notice of Internet Availability of Proxy Materials, to stockholders who have the same address unless the bank, broker or other holder of record has received contrary instructions from one or more of the stockholders. If you wish to receive a separate copy of the Proxy Statement and Annual Report, or Notice of Internet Availability of Proxy Materials, now or in the future, you may contact us at the address or phone number above and we will promptly deliver a separate copy. Beneficial owners sharing an address who are currently receiving multiple copies of the Proxy Statement and Annual Report, or Notice of Internet Availability of Proxy Materials, and wish to receive a single copy in the future, should contact their bank, broker or other holder of record to request that only a single copy be delivered to all stockholders at the shared address in the future.
Who can attend the Annual Meeting?
Only stockholders of Bristol-Myers Squibb as of the record date, March 11, 2010,10, 2011, their authorized representatives and guests of Bristol-Myers Squibb may attend the Annual Meeting. Admission will be by ticket only. A form of government-issued photograph identification will be required to enter the
meeting. Large bags, backpacks, briefcases, cameras, recording equipment and other electronic
devices will not be permitted in the meeting, and attendees will be subject to security inspections. Our offices are wheelchair accessible. We will provide, upon request, wireless headsets for hearing amplification.
How do I receive an admission ticket?
If you are a registered stockholder (your shares are held in your name) and plan to attend the meeting, you should bring either the Notice of Internet Availability of Proxy Materials or the top portion of the proxy card, both of which will serve as your admission ticket.
If you are a beneficial owner (your shares are held in the name of a bank, broker or other holder of record) and plan to attend the meeting, you can obtain an admission ticket in advance by writing to Stockholder Services, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154. Please be sure to enclose proof of ownership, such as a bank or brokerage account statement. Stockholders who do not obtain tickets in advance may obtain them upon verification of ownership at the Registration Desk on the day of the Annual Meeting.
We may also issue tickets to other individuals at our discretion.
Who is entitled to vote?
All holders of record of our $0.10 par value common stock and $2.00 convertible preferred stock at the close of business on March 11, 201010, 2011 will be entitled to vote at the 20102011 Annual Meeting. Each share is entitled to one vote on each matter properly brought before the meeting.
How do I vote if I am a registered stockholder?
Proxies are solicited to give all stockholders who are entitled to vote on the matters that come before the meeting the opportunity to do so whether or not they attend the meeting in person. If you are a registered holder, you can vote your shares by proxy in one of the following manners:
| (i) | via Internet at www.proxyvote.com; |
| (ii) | by telephone at (800) 690-6903; |
| (iii) | by mail, if you received a paper copy of the proxy materials; or |
| (iv) | in person at the Annual Meeting. |
Choosing to vote via Internet or calling the toll-free number listed above will save us expense. In order to vote online or via telephone, have the voting form in hand and either call the number or go to the website and follow the instructions. If you vote via the Internet or by telephone, please do not return a signed proxy card.
If you received a paper copy of the proxy materials and choose to vote by mail, mark your proxy card enclosed with the Proxy Statement, date and sign it, and mail it in the postage-paid envelope.
If you wish to vote in person, you can vote your shares in person at the Annual Meeting.
How do I specify how I want my shares voted?
If you are a registered stockholder, you can specify how you want your shares voted on each proposal by marking the appropriate boxes on the proxy card. The individuals named as proxies on the proxy card will vote your shares in accordance with your instructions. Please review the voting instructions and read the entire text of the proposals and the positions of the Board of Directors in the Proxy Statement prior to marking your vote.
If your proxy card is signed and returned without specifying a vote or an abstention on a proposal, it will be voted according to the recommendation of the Board of Directors on that proposal. That recommendation is shown for each proposal on the proxy card.
How do I vote if I am a beneficial stockholder?
If you are a beneficial stockholder, you have the right to direct your broker or nominee on how to vote the shares. You should complete a voting instruction card which your broker or nominee is obligated to provide you. If you wish to vote in person at the meeting, you must first obtain from the record holder a proxy issued in your name.
Under the rules of the New York Stock Exchange (NYSE), brokers that have not received voting instructions from their customers ten days prior to the meeting date may vote their customers’ shares in the brokers’ discretion on the proposals regarding routine matters, which in most cases includes the ratification of the appointment of the independent registered public accounting firm.
Under NYSE rules, the election of directors, the advisory votes relating to executive compensation and the approval of any management or stockholder proposals are considered “non-discretionary” items, which means that your broker cannot vote your shares on these proposals.
What items will be voted upon at the Annual Meeting?
At the Annual Meeting, we will consider and act on the following items will be voted upon:of business:
| (i) | the election to the Board of Directors the |
| (ii) | the ratification of the appointment of our independent registered public accounting firm; |
| (iii) |
| (iv) | an advisory vote on the frequency of |
| three stockholder proposals, if presented at the meeting. |
We do not know of any other matter that may be brought before the meeting. However, if other matters are properly presented for action, it is the intention of the named proxies to vote on them according to their best judgment.
What are the Board of Directors’ voting recommendations?
For the reasons set forth in more detail later in the Proxy Statement, our Board of Directors recommends a vote FOR the election of each director, FOR the ratification of the appointment of Deloitte & Touche LLP as our independent registered public accounting firm for 2010, FOR eachthe advisory vote on the compensation of our named executive officers, FOR a frequency of every 3 YEARS to hold the three amendments toadvisory vote on the compensation of our Amended and Restated Certificate of Incorporationnamed executive officers and AGAINST each of the three stockholder proposals.
How many votes are needed to have the proposals pass?
Director Elections: A majority of votes cast with respect to each director’s election at the meeting is required to elect each director. A majority of the votes cast means that the number of votes cast “for” a director must exceed the number of votes cast “against” that director in order for the director to be elected. Abstentions will not be counted as votes cast for or against the director and broker non-votes will have no effect on this proposal.
Ratification of our Auditors: The affirmative vote of a majority of the shares present in person or by proxy and entitled to vote on the matter is required for the ratification of the appointment of our independent registered public accounting firm. Broker non-votes will be counted as shares present and entitled to vote. Abstentions will be counted as votes against this proposal and broker non-votes will have no effect on this proposal.
Special Stockholder MeetingsAdvisory Votes: The affirmative vote of the holders of a majority of our outstanding shares present in person or by proxy and entitled to vote on the matter is required to approvefor the Certificate of Amendment to our Amended and Restated Certificate of Incorporation to permit 25%approval of the holdersadvisory vote
on the compensation of our outstanding voting shares to call a special meeting. Abstentionsnamed executive officers and broker non-votes will be counted as votes against this proposal.
Removal of Supermajority Voting Provision – common stockholders: The affirmative vote of the holders of 75% of our outstanding shares is required to approve the Certificate of Amendment to our Amended and Restated Certificate of Incorporation to eliminate the supermajority voting provision applicable to our common stockholders. Abstentions and broker non-votes will be counted as votes against this proposal.
Removal of Supermajority Voting Provisions – preferred stockholders: Both the affirmative vote of the holders of a majority of our outstanding shares entitled toadvisory vote on the matter and the affirmative votefrequency of the holders of at least two-thirdsadvisory vote on the compensation of our outstanding sharesnamed executive officers. Because your votes are advisory, they will not be binding upon our Board of preferred stockDirectors. With respect to the frequency of the advisory vote on the compensation of our named executive officers, our Board may decide that it is requiredin our and our stockholders’ best interests to approvehold such an advisory vote more or less frequently than the Certificate of Amendment toalternative approved by our Amended and Restated Certificate of Incorporation to eliminate the supermajority voting provisions applicable to preferred stockholders. Abstentions and broker non-votes will be counted as votes against the proposal.
Stockholder Proposals: The affirmative vote of the holders of a majority of our outstanding shares present in person or by proxy and entitled to vote on the matter is required for the approval of each of the three stockholder proposals. Abstentions will be counted as votes against these items and broker non-votes will have no effect on this proposal.
How are the votes counted?
In accordance with the laws of Delaware, our Amended and Restated Certificate of Incorporation and our Bylaws, for all matters being submitted to a vote of stockholders, only proxies and ballots that indicate votes “FOR,” “AGAINST” or “ABSTAIN” on the proposals, or in the case of the advisory vote on the frequency of the advisory vote on the executive compensation of our named executive officers, proxies and ballots that indicate “3 YEARS,” “2 YEARS,” “1 YEAR,” or “ABSTAIN” or that provide the designated proxies with the right to vote in their judgment and discretion on the proposals are counted to determine the number of shares present and entitled to vote. Broker non-votes are not counted as shares present and entitled to vote but will be counted for purposes of determining quorum (whether enough votes are present to hold the Annual Meeting).
Can I change my vote after I return the proxy card, or after voting by telephone or electronically?
If you are a stockholder of record, you can revoke your proxy at any time before it is voted at the meeting by taking one of the following three actions:
| (i) | by giving timely written notice of the revocation to the Secretary of Bristol-Myers Squibb; |
| (ii) | by casting a new vote by telephone or by the Internet; or |
| (iii) | by voting in person at the Annual Meeting. |
If you are a beneficial owner of shares, you may submit new voting instructions by contacting your bank, broker or other holder of record. You may also vote in person at the Annual Meeting if you obtain a legal proxy.
All shares that have been properly voted and not revoked will be voted at the Annual Meeting.
How do I designate my proxy?
If you wish to give your proxy to someone other than the Directors’ Proxy Committee, you may do so by crossing out the names of all three Proxy Committee members appearing on the proxy card and inserting the name of another person. The signed card must be presented at the meeting by the person you have designated on the proxy card.
Who counts the votes?
An independent agent tabulates the proxies and the votes cast at the meeting. In addition, independent inspectors of election certify the results of the vote tabulation.
Is my vote confidential?
Yes, any information that identifies a stockholder or the particular vote of a stockholder is kept confidential.
Who will pay for the costs involved in the solicitation of proxies?
We will pay all costs of preparing, assembling, printing and distributing the proxy materials. We have retained Georgeson Shareholder Communications Inc. to assist in soliciting proxies for a fee of $25,000,$18,000, plus reasonable out-of-pocket expenses. We may solicit proxies on behalf of the Board of Directors through the mail, in person, and by telecommunications. We will, upon request, reimburse brokerage firms and others for their reasonable expenses incurred for forwarding solicitation material to beneficial owners of stock.
What is the deadline to propose actions for consideration at next year’s annual meeting of stockholders?
As set forth in our Bylaws, if you wish to propose any action, including the nomination of directors, at next year’s annual meeting, you must deliver notice to BMS containing certain information set forth in our Bylaws, not less than 90 but not more than 120 days before the anniversary of the prior year’s annual meeting. For our 2012 Annual Meeting, we must receive this notice between January 4, 2012 and February 3, 2012. These requirements are separate and distinct from the SEC requirements that a stockholder must meet to have a stockholder proposal included in our proxy statement. For further information on how a stockholder may nominate a candidate to serve as a director, please see page 12.
Our Bylaws are available on our website at www.bms.com/ourcompany/governance. In addition, a copy of the Bylaw provisions discussed above may be obtained by writing to us at our principal executive offices, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Secretary.
What is the deadline to submit stockholder proposals for inclusion in next year’s proxy statement?
Stockholder proposals relating to our 2012 Annual Meeting of Stockholders must be received by us at our principal executive offices, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Secretary, no later than November 23, 2011. Such proposals must comply with SEC regulations under Rule 14a-8 regarding the inclusion of stockholder proposals in company sponsored proxy materials. Stockholders are encouraged to contact the Office of the Corporate Secretary prior to submitting a stockholder proposal or any time they have a concern. At the direction of the Board of Directors, the Office of the Corporate Secretary acts as corporate governance liaison to stockholders.
CORPORATE GOVERNANCE AND BOARD MATTERS
Our business is managed under the direction of our Board of Directors pursuant to the Delaware General Corporation Law and our Bylaws. The Board has responsibility for establishing broad corporate policies and for the overall performance of our company. The Board is kept advised of company business through regular written reports and analyses and discussions with the Chairman and CEO and other officers of Bristol-Myers Squibb, by reviewing materials provided to them and by participating in Board and Board Committee meetings.
The Board of Directors has adopted Corporate Governance Guidelines that govern its operation and that of its Committees. From time to time, our Board revises the Corporate Governance Guidelines in response to changing regulatory requirements, evolving best practices and the concerns of our stockholders and other constituents. Our Corporate Governance Guidelines may be viewed on our website at www.bms.com/ourcompany/governance.
The Committee on Directors and Corporate Governance continually reviews corporate governance issues and is responsible for identifying and recommending the adoption of corporate governance initiatives. At this Annual Meeting, upon the recommendation of the Committee and the Board, we are asking our stockholders to amend our Amended and Restated Certificate of Incorporation to:
eliminate all supermajority voting provisions and
permit stockholders holding at least 25% of our outstanding voting stock to call special meetings of stockholders.
In addition, our Compensation and Management Development Committee regularly reviews compensation issues and recommends adoption of policies and procedures that strengthen our compensation practices. The Compensation Discussion and Analysis beginning on page 2526 discusses many of these policies and procedures.
Listed below are some of the significant corporate governance initiatives we have adopted:
| ||
| Special Stockholder Meetings | • | |
| ||
| Elimination of Supermajority Provisions | • We amended our Amended and Restated Certificate of Incorporation to eliminate the sole supermajority provision applicable to common stockholders. | |
| Director Elections | • Our directors are annually elected by our stockholders.
• Our directors must receive a majority of the votes cast in uncontested elections to be elected.
• We have a director resignation policy that requires a current director to tender his or her resignation to the Board if such director does not receive a majority of the votes cast. The Committee on Directors and Corporate Governance will recommend to the full Board whether to accept the resignation or whether to take other action. | |
| Stockholder Rights Plans | • We do not currently have a stockholder rights
• All stockholder rights plans must be approved by a minimum of two-thirds of the Board.
• All stockholder rights plans must expire one year after Board adoption unless approved by our stockholders. | |
| Related Party Transactions | • We have adopted related party transaction policies and procedures that require the Committee on Directors and Corporate Governance to review and approve related party transactions. These policies and procedures are described in greater detail beginning on page | |
| Political Contributions | • We semi-annually disclose on our website all political contributions to political committees, parties or candidates on both state and federal levels that are made by our company or our employee political action committee. We also disclose the amount of our dues or other payments made to trade associations to which we give $100,000 or more that can be attributed to political contributions.
• Our Committee on Directors and Corporate Governance provides oversight of our political contributions and annually reviews our political contribution policy. | |
The Board of Directors has adopted Corporate Governance Guidelines that govern its operation and that of its Committees. From time to time, our Board revises the Corporate Governance Guidelines in response to changing regulatory requirements, evolving best practices and the concerns of our stockholders and other constituents. Our Corporate Governance Guidelines may be viewed on our website at www.bms.com/ourcompany/governance.
Board’s Role in Strategic Planning and Risk Oversight
Our Board meets regularly to discuss the strategic direction and the issues and opportunities facing our company in light of trends and developments in the biopharmaceutical industry and general business environment. Our Board has been instrumental in determining our strategy to combine the best of biotechnology with pharmaceuticals to become a best-in-class next generation biopharmaceutical company. Throughout the year, our Board provides guidance to management regarding our strategy and helps to refine our operating plans to implement our strategy. Each year, typically during the second quarter, the Board holds an extensive meeting with senior management
dedicated to discussing and reviewing our long-term operating plans and overall corporate strategy. A discussion of key risks to the plans and strategy as well as risk mitigation plans and activities is led by the Chairman andour Chief Executive Officer as part of the meeting. The involvement of the Board in setting our business strategy is critical to the determination of the types and appropriate levels of risk undertaken by the company. As stated in our Corporate Governance Guidelines, our Board is responsible for risk oversight as part of its fiduciary duty of care to effectively monitor business operations. Our Board administers its risk oversight function as a whole and through its Board Committees. For example, the Audit Committee regularly discusses with management our policies and guidelines regarding risk assessment and risk management, as well as our major risk exposures, their potential financial impact on our company and our risk mitigation strategies. In addition, the Audit Committee participates in regular reviews of our process to assess and manage enterprise risk management, including those related to market/environmental, strategic, financial, operational, legal, compliance and reputational risks. In addition, each of the other standing Board committees (the Compensation and Management Development Committee, the Committee on Directors and Corporate Governance, and the Science and Technology Committee), regularly meet to discuss the short-term and long-term objectives and to provide oversight for risks relating to the applicable committee’s areas of responsibility.
It is the policy of our Board that a substantial majority of its members be independent from management, and the Board has adopted independence standards that meet, and in some areas exceed, the listing standards of the New York Stock Exchange. In accordance with our Corporate Governance Guidelines, our Board undertook its annual review of director independence. Our Board considered any and all commercial and charitable relationships of directors, including transactions and relationships between each director or any member of his or her immediate family and Bristol-Myers Squibb and its subsidiaries, which are described more fully below. Following the review, our Board determined, by applying the independence standards contained in the Corporate Governance Guidelines, that each of our directors and each director nominee for election at this Annual Meeting is independent of Bristol-Myers Squibb and its management in that none has a direct or indirect material relationship with our company, except for Lamberto Andreotti, James M. Cornelius and Lamberto Andreotti. Messrs. CorneliusElliott Sigal, M.D., Ph.D. Mr. Andreotti and AndreottiDr. Sigal are not considered independent directors because they are currently employed by our company. In addition, Mr. Cornelius is not considered independent because of Mr. Cornelius’his previous employment, within the past three years, as Chief Executive Officer and Mr. Andreotti’s employment as President, Chief Operating Officer and CEO-Designate of our company.
In determining that each of Lewis B. Campbell, Louis J. Freeh, Laurie H. Glimcher, M.D., Michael Grobstein, Leif Johansson, Alan J. Lacy, Vicki L. Sato, Ph.D., Togo D. West, Jr. and R. Sanders Williams, M.D. is independent, the Board considered the following relationships under our categorical standards (see Exhibit A):
Drs. Glimcher Sato and Williams and Mr. LacySato are directors or trustees of companies that received payment from the company for property or services in an aggregate amount whichthat did not exceed the greater of $1 million or 2% of such other company’s consolidated gross revenues. TheFor each transaction, the Board determined that for each such transaction, the director did not initiate or negotiate the transaction and that the transaction was entered into in the ordinary course of business.
Drs. Glimcher, Sato and Williams, or one of their immediate family members, are or were employed by educational or medical institutions with which we engage in ordinary course business transactions. The director did not initiate or negotiate any transaction with such institution and the payments made did not exceed the greater of $1 million or 2% of such institution’s consolidated gross revenues.
business transactions. The director did not initiate or negotiate any transaction with such institution and the payments made did not exceed the greater of $1 million or 2% of such institution’s consolidated gross revenues. |
Mr. West is a trusteeGrobstein and Dr. Sato are directors of a charitable organization or non-profit organizationorganizations to which the company, or the Bristol-Myers Squibb Foundation made a charitable contribution,contributions, which, in the aggregate, did not exceed the greater of $1 million or 2% of that organization’s consolidated gross revenues.
Our CEO, James M. Cornelius, also currently serves as the Chairman of the Board of Directors. Our Board determined that it was appropriate to combine the roles of Chairman and CEO to help guide the company through its transition to a next generation biopharmaceutical company. As previously announced,On May 4, 2010, Mr. Cornelius will retireretired as our CEO on May 4, 2010 and becomebecame our Non-Executive Chairman. LambertoChairman and Mr. Andreotti will becomebecame our new CEO on that date.CEO. The Board has determined at that time that it iswas appropriate to separate the roles of the Chairman and the CEO at this time in view of the completion of our transformation into a next generation biopharmaceutical company and Mr. Cornelius’sCornelius’ retirement as CEO. Mr. Cornelius will coach and mentor Mr. Andreotti as he transitions into his new role as CEO. The Board has determined to maintain a Lead Independent Director at this time because Mr. Cornelius does not meet the New York Stock Exchange standards of independence due to his prior service as Chief Executive Officer of the company. The independent directors have elected Lewis B. Campbell, the current Chair of the Compensation and Management Development Committee, to serve as the Lead Independent Director. The Lead Independent Director is selected annually by the independent directors. The Lead Independent Director’s responsibilities include, among others, presiding at the meetings of independent directors, approving meeting agendas and meeting schedules, approving and advising the Chairman as to the quality, quantity and timeliness of information sent to the Board and serving as the principal liaison and facilitator between the independent directors and the Chairman. A more detailed description of the roles and responsibilities of the Lead Independent Director is available on our website at www.bms.com/ourcompany/governance.
Our Board meets on a regularly scheduled basis during the year to review significant developments affecting Bristol-Myers Squibb and to act on matters requiring Board approval. It also holds special meetings when important matters require Board action between scheduled meetings. Members of senior management regularly attend Board meetings to report on and discuss their areas of responsibility. In 2009,2010, the Board of Directors met 1310 times. The average aggregate attendance of directors at Board and committee meetings was over 93%98%. No director attended fewer than 75% of the aggregate number of Board and committee meetings during the period he or she served. In addition, our independent directors met in non-management director sessions six times during 20092010 to discuss such topics as our independent directors determined, including the evaluation of the performance of our Chief Executive Officer. Mr. Campbell, our Lead Independent Director, presided over these sessions.
Annual Meeting of Stockholders
All of the 2009 nominees for director attended our 2009 Annual Meeting of Stockholders. Directors are strongly encouraged, but not required, to attend the Annual Meeting of Stockholders. All of the 2011 nominees for director attended our 2010 Annual Meeting of Stockholders, except for Dr. Glimcher who, for the first time in over 14 years, could not attend our Annual Meeting due to a prior long-standing commitment.
Our Bylaws specifically provide for an Audit Committee, Compensation and Management Development Committee, and Committee on Directors and Corporate Governance, which are composed entirely of independent directors. Our Board of Directors has determined, in its judgment, that all members of the Audit Committee are financially literate and that all members of the Audit Committee meet additional, heightened independence criteria applicable to directors serving on audit committees under the New York Stock Exchange listing standards. In addition, our Board has determined that Michael Grobstein and Alan J. Lacy each qualify as an “audit committee financial expert” under the applicable SEC rules.
Our Bylaws also authorize the establishment of additional committees of the Board and, under this authorization, our Board of Directors established the Science and Technology Committee. Our Board has appointed individuals from among its members to serve on these four committees and each
committee operates under a written charter adopted by the Board, as amended from time to time. These charters are published on our website at www.bms.com/ourcompany/governance. Each of these Board committees has the necessary resources and authority appropriate to discharge its responsibilities, including the authority to retain consultants or experts to advise the committee.
On January 20, 2009, the Board established an IPO Committee to determine and approve the terms of the partial initial public offering of Mead Johnson Nutrition Company. The members of the IPO Committee were Messrs. Grobstein, Cornelius and Campbell. The IPO Committee met twice and upon the completion of the partial IPO of Mead Johnson, the Committee was dissolved.
The table below indicates the current members of the Audit Committee, Compensation and Management Development Committee, Committee on Directors and Corporate Governance and Science and Technology Committee, the primary responsibilities of each committee and the number of meetings held in 2009:2010:
| Committee | Current Members | Primary Responsibilities | No. of Meetings | |||
Audit | Michael Grobstein (Chair) Lewis B. Campbell Louis J. Freeh Laurie H. Glimcher, M.D. Leif Johansson Alan J. Lacy | • overseeing and monitoring the quality of our accounting and auditing practices; • appointing, compensating and providing oversight of the performance of our independent registered public accounting firm for the purpose of preparing or issuing audit reports and related work regarding our financial statements and the effectiveness of our internal control over financial reporting; • assisting the Board in fulfilling its responsibilities for general oversight of (i) compliance with legal and regulatory requirements, (ii) the performance of our internal audit function and • reviewing our disclosure controls and procedures, including internal controls, periodic filings with the U.S. Securities and Exchange Commission (SEC), earnings releases and earnings guidance; • producing the required Audit Committee Report for inclusion in our Proxy Statement; and • overseeing investigations into complaints concerning financial or accounting matters. | ||||
| Committee | Current Members | Primary Responsibilities | No. of Meetings | |||
Compen- sation and Management Development | Lewis B. Campbell (Chair) Michael Grobstein Leif Johansson Vicki L. Sato, Ph.D. Togo D. West, Jr. | • reviewing, approving and reporting to our Board on our major compensation and benefits plans, policies and programs; • annually reviewing and approving corporate goals and objectives relevant to CEO compensation, evaluating the CEO’s performance in light of those goals and objectives and recommending for approval by three-fourths of our independent directors the CEO’s compensation based on this evaluation; • reviewing and evaluating the performance of senior management; approving the compensation of executive officers and certain senior management; • overseeing our management development programs, performance assessment of senior executives and succession planning; • reviewing and discussing with management the Compensation Discussion and Analysis and related disclosures required for inclusion in our Proxy Statement, recommending to the Board, whether the Compensation Discussion and Analysis should be included in our Proxy Statement, and producing the Compensation and Management Development Committee Report required for inclusion in our Proxy • reviewing incentive compensation programs to confirm incentive pay does not encourage unnecessary risk-taking. | ||||
Directors and Corporate Governance | Louis J. Freeh (Chair) Lewis B. Campbell Laurie H. Glimcher, M.D. Alan J. Lacy Togo D. West, Jr. R. Sanders Williams, M.D. | • developing and recommending to our Board a set of Corporate Governance Guidelines and periodically reviewing such guidelines; • identifying and recommending corporate governance best practices; • reviewing and recommending annually to our Board of Directors the compensation of non-employee directors; • considering questions of potential conflicts of interest of directors and senior management, including approving related party transactions; • defining specific categorical standards for director independence; • considering matters of corporate social and public responsibility and matters of significance in areas related to corporate public affairs and our employees and stockholders; • identifying individuals qualified to become Board members and recommending that our Board select the director nominees for the next annual meeting of stockholders; and • overseeing our Board’s annual evaluation of its performance. | ||||
| Committee | Current Members | Primary Responsibilities | No. of Meetings | |||
| Science and Technology | Laurie H. Glimcher, M.D. (Chair) Vicki L. Sato, Ph.D. R. Sanders Williams, M.D. Lamberto Andreotti Elliott Sigal, M.D., Ph.D. | • reviewing and advising our Board on the strategic direction of our research and development (R&D) programs and our progress in achieving long-term R&D objectives; • reviewing and advising our Board on our internal and external investments in science and technology; and • identifying and discussing significant emerging trends and issues in science and technology and considering their potential impact on our company. | 5 | |||
Compensation Committee Interlocks and Insider Participation
There were no Compensation and Management Development Committee interlocks or insider (employee) participation in 2009.2010.
Risk Assessment of Compensation Policies and Practices
We have reviewedIn 2010, we conducted a worldwide review of our material compensation policies and practices and have concluded that these policies and practices are not reasonably likely to have a material adverse effect on the company. Specifically,On a global basis, our compensation programs contain many design features that mitigate the likelihood of inducing excessive risk-taking behavior. These features include:
a balance of fixed and variable compensation, with variable compensation tied both to short-term objectives and the long-term value of our stock price;
multiple metrics in our incentive programs that balance top-line, bottom-line and cash management objectives;
linear payout curves and caps in our incentive program payout formulas;
reasonable goals and objectives in our incentive programs;
payouts modified based upon individual performance, inclusive of assessments against the our Core BMS Behaviors and the BMS Commitment;
the Compensation and Management Development Committee’s ability to exercise downward discretion in determining incentive program payouts;
clawback provisions pertaining to annual incentive payouts and long-term incentive awards;
share ownership and retention guidelines applicable to our top 50senior executives;
equity award policies that limit risk by not allowing for timing of equity award grants;
prohibition of speculative and hedging transactions by all employees;
all managers and executives worldwide participate in the same annual incentive program that pertains to our Named Executive Officers and that has been approved by the Compensation and Management Development Committee; and
mandatory training on our Standards of Business Conduct and Ethics and other policies that educate our employees on appropriate behaviors and the consequences of taking inappropriate actions.
Our Corporate Governance Guidelines contain Board membership criteria that apply to nominees for a position on our Board of Directors. Under these criteria, members of our Board should be persons
of diverse backgrounds with broad experience in areas important to the operation of our company such as business, science, medicine, finance/accounting, law, education or government and should possess qualities reflecting integrity, independence, wisdom, an inquiring mind, vision, a proven record of accomplishment and an ability to work well with others. In addition, each director must represent the interests of our stockholders. We do not have a formal policy on Board diversity as it relates to race, gender or national origin.
Identification and Selection of Nominees for our Board
The Committee on Directors and Corporate Governance periodically assesses the appropriate size of our Board, and whether any vacancies on our Board are expected due to retirement or otherwise. In the event that vacancies are anticipated or otherwise arise, the Committee on Directors and Corporate Governance considers candidates for Board membership. Candidates may come to the attention of the Committee on Directors and Corporate Governance through current Board members, third-party search firms, management, stockholders or others. The Chair of the Committee on Directors and Corporate Governance, in consultation with the Chairman, and CEO, conducts an initial evaluation of the prospective nominees against the established Board membership criteria discussed above. The Committee reviews the skills of the current directors and compares them againstto the particular skills of potential candidates, keeping in mind its commitment to maintain a Board with members of diverse experience. Additional information relevant to the qualifications of prospective nominees may be requested from third-party search firms, other directors, management or other sources. After this initial evaluation, prospective nominees may be interviewed by telephone or in person by the Chair of the Committee on Directors and Corporate Governance, the Chairman and CEO and other directors. After completing this evaluation and interview, the Committee on Directors and Corporate Governance makes a recommendation to the full Board as to the persons who should be nominated by our Board, and the full Board determines the nominees after considering the recommendation and any additional information it may deem appropriate.
Stockholder Nominations for Director
The Committee on Directors and Corporate Governance considers and evaluates stockholder recommendations of nominees for election to our Board of Directors in the same manner as other director nominees. Stockholder recommendations must be accompanied by a comprehensivedisclosure including written resume ofinformation about the recommended nominee’s business experience and background with a consent in writing signed by the recommended nominee that he or she is willing to be considered as a nominee and, if nominated and elected, he or she will serve as a director. Stockholders should send their written recommendations of nominees accompanied by the aforesaidrequired documents to the principal executive offices of the company addressed to: Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Secretary.
Our Board of Directors has nominated eleventwelve current directors, Lamberto Andreotti, Lewis B. Campbell, James M. Cornelius, Louis J. Freeh, Laurie H. Glimcher, M.D., Michael Grobstein, Leif Johansson, Alan J. Lacy, Vicki L. Sato, Ph. D.Ph.D., Elliott Sigal, M.D., Ph.D., Togo D. West, Jr. and R. Sanders Williams, M.D., to serve as directors of Bristol-Myers Squibb. The directors will hold office from election until the 20112012 Annual Meeting.
A majority of the votes cast is required to elect directors. Any current director who does not receive a majority of votes cast must tender his or her resignation as a director within 10 business days after the certification of the stockholder vote. The Committee on Directors and Corporate Governance, without participation by any director tendering his or her resignation, will consider the resignation offer and recommend to the Board whether to accept it. The Board, without participation by any director tendering his or her resignation, will act on the Committee’s recommendation at its next regularly scheduled meeting to be held within 60 days after the certification of the stockholder vote. We will promptly disclose the Board’s decision and the reasons for that decision in a broadly disseminated press release that will also be furnished to the Securities and Exchange Commission on Form 8-K. If any nominee is unable to serve, proxies will be voted in favor of the remainder of those nominated and may be voted for substitute nominees, unless our Board of Directors provides for a lesser number of directors.
Listed below is certain biographical information of each of the nominees for election including his or her principal occupation, current directorships and directorships held during the past 5 years of public companies and registered investment advisors and other business affiliations. Also included is a description of the specific experience, qualifications, attributes and skills of each nominee that led the Board to conclude that each nominee is well-qualified to serve as a member of our Board of Directors.
Information on Nominees for Directors
Director since 2009 | LAMBERTO ANDREOTTI
Mr. Andreotti, age
With his
During the last 5 years, Mr. Andreotti | |
Director since 1998 | LEWIS B. CAMPBELL
Mr. Campbell, age
Mr. Campbell is a demonstrated leader with keen business understanding. With his focus on operational efficiencies at Textron, Mr. Campbell is uniquely positioned to help guide the company through its transition to a biopharmaceutical company. Furthermore, his first-hand knowledge of the many issues facing public companies and his service on all of our independent Board Committees position him well to serve as our Lead Independent Director.
During the last 5 years, Mr. Campbell | |
Director since 2005 | JAMES M. CORNELIUS
Mr. Cornelius, age
Mr. Cornelius has been the principal strategist in the company’s transition into a next generation biopharmaceutical company. His extensive understanding of the complex operational, regulatory and financial issues facing pharmaceutical companies and general industry gained while serving in key leadership roles at Eli Lilly and Co. and Guidant Corporation and on the boards of other companies positions Mr. Cornelius well to serve as the Non-Executive Chairman of our Board of Directors.
Mr. Cornelius is the Chairman of the Board of Directors of Mead Johnson Nutrition Company and a Director of Given Imaging Ltd. During the last 5 years, Mr. Cornelius was | |
Director since 2005 | LOUIS J. FREEH
Mr. Freeh, age
Mr. Freeh brings a unique perspective to our Board based on his extensive experience as a former federal judge, FBI director and corporate general counsel, particularly with respect to matters relating to law, corporate governance, compliance and regulatory matters. He has a demonstrated record of integrity and independence.
Mr. Freeh is a Director of Wilmington | |
Director since 1997 | LAURIE H. GLIMCHER, M.D.
Dr. Glimcher, age
Dr. Glimcher is an internationally known immunologist and physician who brings a unique perspective to our Board on a variety of healthcare related issues. She is also a Senior Physician and Rheumatologist at Brigham and Women’s Hospital. Dr. Glimcher serves on the Memorial Sloan-Kettering Cancer Center Board of Scientific Consultants and on the Scientific Advisory Boards of the Burroughs-Wellcome Fund, Cancer Research Institute, Immune Disease Institute, Health Care Ventures, Inc., Nodality Inc., Abpro, Inc., Theraclone Sciences, Inc. and American Asthma Foundation. She is also a member of the American Association of Physicians and the Institutes of Medicine of the National Academy of Sciences. Her expertise in the immunology area and her extensive experience in the medical field position her well to serve as the Chair of the Science and Technology Committee.
Dr. Glimcher is a Director of Waters | ||
Director since 2007 | MICHAEL GROBSTEIN
Mr. Grobstein, age
With over 30 years experience at a major auditing firm, Mr. Grobstein has extensive knowledge and background relating to accounting and financial reporting rules and regulations as well as the evaluation of financial results, internal controls and business processes. Mr. Grobstein’s depth and breadth of financial expertise and his experience handling complex financial issues as chair of the audit committees of two other public companies in the healthcare industry position him well to serve as Chair of our Audit Committee.
Mr. Grobstein is a Director of Given Imaging Ltd. | |
Director since 1998 | LEIF JOHANSSON
Mr. Johansson, age
Mr. Johansson is a highly respected global business leader who provides a wealth of management experience and business expertise. Mr. Johansson has a comprehensive understanding of the operational, financial and strategic challenges facing global companies and knows how to make businesses work effectively and efficiently.
Mr. Johansson is a Director of AB Volvo and Svenska Cellulosa Aktiebolaget SCA. | ||
Director since 2008 | ALAN J. LACY
Mr. Lacy, age
Mr. Lacy brings to the Board extensive business understanding and demonstrated management expertise having served in key leadership positions at Sears Holdings Corporation, including Chief Executive Officer. In addition, his experience as a senior financial officer of three large public companies provides him with a comprehensive understanding of the complex financial and legal issues facing large companies and makes him a key member of the Audit Committee. Mr. Lacy is a highly respected business leader with a proven record of accomplishment.
Mr. Lacy is a Director of The Western Union Company, The Hillman Companies, Inc. and Dave & Buster’s, Inc. He is also a Trustee of Fidelity Funds. | |
Director since 2006 | VICKI L. SATO, PH.D.
Dr. Sato, age
Dr. Sato’s extensive and distinctive experience in business, academia and science over more than 25 years brings to the Board a valuable perspective on the biotech industry. Dr. Sato has a strong background in research and development positioning her well to serve on the Science and Technology Committee. Her service as chair of the compensation committees of two healthcare companies makes Dr. Sato a well-qualified member of our Compensation and Management Development Committee.
Dr. Sato is a Director of PerkinElmer Corporation and Alnylam Pharmaceuticals. | |
Director since 2011 | ELLIOTT SIGAL, M.D., PH.D.
Dr. Sigal, age 59, has been our Chief Scientific Officer and President of Research and Development since October 2004 and an Executive Vice President since October 2006. Dr. Sigal joined BMS in November 1997 as Vice President of the newly created department of Applied Genomics and since then has held positions of increasing responsibility in both discovery and development. Dr. Sigal has more than 25 years of combined experience in medicine, research and management and is highly respected by the biopharmaceutical industry. Dr. Sigal serves a key role as a member of the Board’s Science and Technology Committee as the continued importance of research and development is critical to our strategy and our success as a biopharma company. During the last 5 years, Dr. Sigal was a Director of Mead Johnson Nutrition Company. | |
Director since 2008 | TOGO D. WEST, JR. Secretary West, age
Secretary West’s government experience provides the Board with a unique perspective of the issues facing our company. In his position as Secretary of Veterans Affairs, he was a member of the President’s Cabinet, and oversaw the largest healthcare system in the country; and as Secretary of the Army, he was responsible for all Army activities, including the extensive system of Army medical centers around the world. In 2007, Secretary West was asked to co-chair the review of the delivery of healthcare at Walter Reed Army Medical Center and the National Naval Medical Center at Bethesda. With his keen understanding of the public policy issues facing the healthcare industry, Secretary West is a key member of the Committee on Directors and Corporate Governance.
Secretary West is a Director of FuelCell Energy, Inc., and Krispy Kreme | |
Director since 2006 | R. SANDERS WILLIAMS, M.D.
Dr. Williams, age 61, has been President and Robert W. and Linda L. Mahley Distinguished Professor of The J. David Gladstone Institutes, a research facility and Professor of Medicine at the University of California, San Francisco, since March 2010. From 2007 to 2010, Dr. Williams was the Senior Vice Chancellor for Academic Affairs at Duke University Medical Center and Dean of Duke University School of Medicine from 2001 to 2007. Dr. Williams joined the Duke faculty in 1980 as an assistant professor of medicine, physiology and cell biology. Dr. Williams is a consultant to Phrixus, Inc and the Taylor Companies.
Dr. Williams has served recently on the Director’s Advisory Committee of the National Institutes of Health and the Board of External Advisors to the National Heart, Lung and Blood Institute. Dr. Williams is also a member of the Institutes of Medicine of the National Academy of Sciences and a fellow of the American Association for the Advancement of Science. With his medical expertise, leadership of clinical care programs and research experience in biology, Dr. Williams brings a valuable perspective to our Board and is a key member of the Science and Technology Committee. In addition, his experience as a practicing physician provides unique insight into the challenges facing patients.
Dr. Williams is a Director of Laboratory Corporation of America. | |
Communications with our Board of Directors
The Committee on Directors and Corporate Governance has created a process by which an interested party may communicate directly with our non-management directors. Any interested party wishing to contact a non-management director may do so in writing by sending a letter to:
[Name of Director]
c/o Corporate Secretary
Bristol-Myers Squibb Company
345 Park Avenue
New York, NY 10154
Any matter relating to our financial statements, accounting practices or internal controls should be addressed to the Chair of the Audit Committee. All other matters should be addressed to the Chair of the Committee on Directors and Corporate Governance. Our Corporate Secretary reviews all correspondence and regularly forwards to our Board a summary of all such correspondence and copies of all correspondence that, in the opinion of our Corporate Secretary, deals with the functions of our Board or its committees, or that our Corporate Secretary otherwise determines requires Board attention. Directors may at any time review a log of the correspondence we receive that is addressed to members of the Board and request copies of any such correspondence.
Our Board of Directors has adopted the Standards of Business Conduct and Ethics that sets forth important company policies and procedures in conducting our business in a legal, ethical and responsible manner. These standards are applicable to all of our employees, including the Chief Executive Officer, the Chief Financial Officer and the Controller. In addition, the Audit Committee has adopted the Code of Ethics for Senior Financial Officers that supplements the Standards of Business Conduct and Ethics by providing more specific requirements and guidance on certain topics. The Code of Ethics for Senior Financial Officers applies to the Chief Executive Officer, the Chief Financial Officer,
the Controller, the Treasurer and the heads of major operating units. Our Board has also adopted the Code of Business Conduct and Ethics for Directors that applies to all directors and sets forth guidance
with respect to recognizing and handling areas of ethical issues. The Standards of Business Conduct and Ethics, the Code of Ethics for Senior Financial Officers and the Code of Business Conduct and Ethics for Directors are available on our website at www.bms.com/ourcompany/governance. We will post any substantive amendments to, or waivers from, applicable provisions of our Standards of Business Conduct and Ethics, our Code of Ethics for Senior Financial Officers, and our Code of Business Conduct and Ethics for Directors on our website at www.bms.com/ourcompany/governance within two days following the date of such amendment or waiver.
Employees are required to report any conduct they believe in good faith to be an actual or apparent violation of our Codes of Conduct. In addition, as required under the Sarbanes-Oxley Act of 2002, the Audit Committee has established procedures to receive, retain and treat complaints received regarding accounting, internal accounting controls, or auditing matters and the confidential, anonymous submission by company employees of concerns regarding questionable accounting or auditing matters.
The Board has adopted written policies and procedures for the review and approval of transactions involving the company and related parties, such as directors, executive officers and their immediate family members. The policy covers any transaction or series of transactions (an “interested transaction”) in which the amount involved exceeds $120,000, the company is a participant, and a related party has a direct or indirect material interest (other than solely as a result of being a director or less than 10 percent beneficial owner of another entity). All interested transactions are subject to approval or ratification in accordance with the following procedures:
Management will be responsible for determining whether a transaction is an interested transaction requiring review under this policy, in which case the transaction shall be disclosed to the Committee on Directors and Corporate Governance.
The Committee on Directors and Corporate Governance shall review the relevant facts and circumstances, including, among other things, whether the interested transaction is on terms no less favorable than terms generally available to an unaffiliated third party under the same or ordinary circumstances and the related party’s interest in the transaction.
If it is impractical or undesirable to wait until a Committee meeting to consummatecomplete an interested transaction, the Chair of the Committee in consultation with the General Counsel may review and approve the transaction, which approval must be ratified by the Committee at its next meeting.
In the event the company becomes aware of an interested transaction that has not been approved, the Committee shall evaluate all options available to the company, including ratification, revision or termination of such transaction and take such course of action as the Committee deems appropriate under the circumstances.
No director shall participate in any discussion or approval of the interested transaction for which he or she is a related party.
If an interested transaction is ongoing, the Committee may establish guidelines for management to follow in its ongoing dealings with the related party and shall review and assess such ongoing relationships on at least an annual basis.
Certain types of interested transactions are deemed to be pre-approved or ratified by the Committee, as applicable, including the employment of executive officers, director compensation, certain transactions with other companies or charitable contributions, transactions where all shareholders receive proportional benefits, transactions involving competitive bids, regulated transactions and certain banking-related services.
The Committee on Directors and Corporate Governance has approved the following related party transaction in accordance with our policy and Bylaws:
The Officers and Directors Indemnification Trust One was formed on October 20, 2005 pursuant to the settlement agreement among Bristol-Myers Squibb Company, our directors and officers, and our D&O insurers. The trust was formed to fund, under certain conditions, the payment of any settlement of judgment costs, including any award of attorneys’ fees to counsel for plaintiffs, incurred by or on behalf of certain of our current and former directors and officers in any derivative lawsuit brought on behalf of Bristol-Myers Squibb for which a claim for coverage under the policies released in the settlement could have been made. Insurance proceeds received in the settlement in the amount of $38.5 million have beenwere deposited into this trust. An independent Trustee administersadministered the Trust. NoTrust and no distribution was made in 2009. If2010. Pursuant to the Trust has not already been terminated pursuant toterms of the Trust Agreement, it will terminatethe Trust was terminated on October 20, 2010. Bristol-Myers Squibb, isas the residual beneficiary of the Trust, and will therefore receive anyreceived all trust property that remainsremained in the Trust upon its termination.
Availability of Corporate Governance Documents
Our Corporate Governance Guidelines (including the standards of director independence), Standards of Business Conduct and Ethics, Code of Ethics for Senior Financial Officers, Code of Business Conduct and Ethics for Directors, additional policies and guidelines, committee charters, links to Reports of Insider Transactions and listings of our Board of Directors and Executive Officers are available on our corporate governance webpage at www.bms.com/ourcompany/governance and are available to any interested party who requests them by writing to: Secretary, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154.
20092010 Director Compensation Program
We aim to provide a competitive compensation program to attract and retain high quality directors. The Committee on Directors and Corporate Governance annually reviews our directors’ compensation practices and compares them against the practices of the companies in our peer group. Our peer group is listed on page 30. The Committee submits its recommendations for director compensation to the full Board for approval. In December 2009, when the directors approved the 2010 compensation for directors, the following companies were in our peer group: Abbott Laboratories, Amgen, Eli Lilly & Co., Johnson & Johnson, Merck & Co., Pfizer, Schering-Plough and Wyeth. Mr. Andreotti did not receive any additional compensation for serving as a director in 2010. Mr. Cornelius received compensation for his services as Non-Executive Chairman commencing on May 4, 2010.
Management has engaged an outside consultant, Frederic W. Cook & Co., Inc. (FWC), to review market data and competitive information on director compensation. Consistent with our desire to attract and retain highly skilled and experienced directors, the Committee on Directors and Corporate Governance, in consultation with FWC, determined that it was appropriate to target director compensation betweenat the median and 75% of the companies in our peer group. In addition, FWC recommended simplifying our director compensation program by eliminating per meeting fees and introducing committee membership retainers. Because the company’s nine non-management directors isare fewer than the peer median of 11, the aggregate cost of our director compensation program is between the peer group 25th percentile and median. Messrs. Andreotti and Cornelius did not receive any additional compensation for serving as a director. The Committee believes the total compensation package for directors we offer continues to be reasonable, and appropriately aligns the interests of directors to stockholders by ensuring directors have a proprietary stake in our company.
The components of our standard non-management directors’ compensation for 20092010 were as follows:
Cash Compensation
In 2009,2010, our non-management directors received an annualwere entitled to receive the following cash compensation:
Annual cash retainer of $60,000 and an additional fee$85,000;
Annual Committee Chair cash retainer of $2,000$25,000 for attending each Board meeting, Board Committee meeting, and the Annual Meeting of Stockholders. In addition, the Lead Independent Director and each of the Chairs of the Audit Committee, the Compensation and Management Development Committee, the Committee on Directors and Corporate Governance and the Science and Technology Committee;
Annual Committee membership cash retainer of $15,000 for each director serving as a member (but not Chair) of the Audit, Compensation and Management Development, and Science and Technology Committees; and
Annual Committee membership cash retainer of $7,500 for each director serving as a member (but not Chair) of the Committee on Directors and Corporate Governance.
In addition, the Lead Independent Director received an annual cash retainer of $20,000.
Deferral Program
A non-management director may elect to defer payment of all or part of the cash compensation received as a director under our company’s 1987 Deferred Compensation Plan for Non-Employee Directors. The election to defer is made in the preceding calendar year in which the compensation is earned. Deferred funds may be credited to one or more of the following funds: a 6-month United States Treasury bill equivalent fund, a fund based on the return on the company’s invested cash or a fund based on the return on our common stock. Deferred portions are payable in a lump sum or in a maximum of ten annual installments. Payments under the Plan begin when a participant ceases to be a director or at a future date previously specified by the director. In December 2009, the 1987 Deferred Compensation Plan for Non-Employee Directors was amended to requireFor all futurecurrent directors, deferred share units, including the annual award, to beare settleable solely in shares of company common stock.
Equity Compensation
Under the 1987 Deferred Compensation Plan for Non-Employee Directors, on February 1, 2009,2010, all non-management directors received an annual award of deferred common share units valued at $110,000. These deferred common share units are non-forfeitable at grant. In addition, ournew non-management directors determined that it was more equitable to grant new directorsreceive a pro-rata portion of the annual deferred share unit award at the time the director joins the company’s Board.
Share Retention Requirements
In 2009,2010, all non-management directors were required to acquire 5,000hold at least $300,000 worth of BMS shares and/or share units within three years of joining the Board and maintain this ownership level while servingthroughout their service as a director. We required that 25% of the annual retainer be deferred and credited to a deferred compensation account, the value of which is determined by the value of our common stock, until a non-management director has attained our share retention requirements. All of our current directors have met our share retention requirements.
Charitable Contribution Programs
Each current director participates in our Directors’ Charitable Contribution Program. Upon the death of a director, we will donate up to an aggregate of $500,000 to up to five qualifying charitable organizations designated by the director. Individual directors derive no financial or tax benefit from this program since the tax benefit of all charitable deductions relating to the contributions accrues solely to us. In 2009, the Board eliminated the Charitable Contributions Program for all new directors.
Each director was also able to participate in our company-wide matching gift program in 2009.2010. We matched dollar for dollar a director’s contribution to qualified charitable and educational organizations
up to $20,000. This benefit was also available to all company employees. In 2009,2010, each of the following directors participated in our matching gift programs as indicated in the Director Compensation Table below: Messrs. Campbell, Freeh, Grobstein and Lacy and Drs. Glimcher, Sato and Williams.
2010 Director Compensation
In December 2009, the Committee on Directors and Corporate Governance reviewed market data and other analyses prepared by FWC. FWC recommended that the directors continue to be positioned at between the 50th and 75th percentile amongst peers, and that no significant changes to the compensation levels be made at this time. To simplify our directors compensation program, FWC recommended eliminating per meeting fees and introducing committee membership retainers. Based on this analysis, the Committee recommended, and the Board approved, effective January 1, 2010, that the non-employee directors be compensated as follows:
Annual Director cash retainer of $85,000;
Annual Committee Chair cash retainer of $25,000 for each of the Chairs of the Audit Committee, Compensation and Management Development Committee, Committee on Directors and Corporate Governance and Science & Technology Committee;
Annual Committee membership cash retainer of $15,000 for each director serving as a member (but not Chair) of the Audit, Compensation and Management Development, and Science and Technology Committees;
Annual Committee membership cash retainer of $7,500 for each director serving as a member (but not Chair) of the Committee on Directors and Corporate Governance; and
An annual grant of deferred share units equal to $110,000 that will be payable on February 1 of each year.
The Board also approved modifications to the Board’s share retention guidelines to denominate the ownership threshold in a dollar value and to require directors to retain a greater value in shares. Directors are now required to hold at least $300,000 worth of company stock or stock units. All of our current directors have met our share retention requirements. In addition, all future grants of deferred share units will be settleable solely in shares of common stock as opposed to cash, which is better aligned with stockholders’ interests. The Board also eliminated the Charitable Contributions Program that provided for a death benefit to one or more charitable organizations for all new directors. Thus, the only perquisite that is offered to directors (which is also offered to all employees of the company) is participation in the company’s matching gift program.
Compensation of the Non-Executive Chairman
On May 4, 2010, Mr. Cornelius will becomebecame our Non-Executive Chairman of the Board. As Non-Executive Chairman, Mr. Cornelius will havehas significantly greater responsibilities than other directors, including chairing the Office of the Chairman to meet on a regular basis with the CEO on the most critical strategic issues and transactions, serving as a liaison between the CEO and the independent directors, frequently discussing the strategy and direction of the company with senior management, and serving as a non-voting member, ex-officio, of the Audit Committee, Committee on Directors and Corporate Governance and the Compensation and Management Development Committee. In addition to the standard Board compensation that all non-employee directors receive, the Board approved, based on a recommendation by FWC, an annual Non-Executive Chairman retainer of $200,000, paid quarterly, of which 50% will beis paid in cash and 50% in shares of company common stock. The Board also recognized that during a transition period, Mr. Cornelius willwould need to spend additional time working with the new CEO on transitional and other company matters. For these services, upon consultation with FWC, the Board approved an annual Transitional Non-Executive Chairman retainer of $175,000, paid quarterly, of which 50% will bewas paid in cash and 50% in shares of company common stock. Mr. Cornelius willwas also be reimbursed for business travel and for office space, utilities, supplies and administrative support for his work related to company matters.
2011 Director Compensation
In December 2010, the Committee on Directors and Corporate Governance reviewed market data and other analyses prepared by FWC that compared our director compensation program against an expanded peer group that included the addition of three biotechnology companies. The companies in this peer group are the same as the companies in the primary peer group for executive compensation purposes and are listed on page 31. With the addition of the three new companies, our director compensation program was positioned at below the 25th percentile. FWC recommended that the directors continue to be positioned at median amongst our peers. Therefore, to bring our directors compensation program to median, FWC recommended, and the Board approved, effective January 1, 2011, that the non-employee directors’ compensation be modified as follows:
Increasing the annual Committee Chair cash retainer to $30,000 for each of the Chairs of the Audit Committee, Compensation and Management Development Committee, Committee on Directors and Corporate Governance and Science & Technology Committee;
Increasing the Lead Director annual retainer to $30,000; and
Increasing the annual grant of deferred share units to $125,000 in value.
In addition, the Committee recommended, and the Board approved, the elimination of the Transitional Non-Executive Chairman Retainer for Mr. Cornelius after December 31, 2010.
Director Compensation Table
The following table sets forth information regarding the compensation earned by our non-employee directors in 2009:2010:
Name | Fees Earned or Paid in Cash(1) | Stock Awards(2) | Option Awards(3) | Change in Pension Value and Non-Qualified Deferred Compensation Earnings(4) | All Other Compensation(5) | Total | Fees Earned or Paid in Cash(1) | Stock Awards(2) | Option Awards(3) | Change in Pension Value and Non-Qualified Deferred Compensation Earnings(4) | All Other Compensation(5) | Total | ||||||||||||||||||||||||||||||
L. Andreotti(6) | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | ||||||||||||||||||||||||||||||
L. B. Campbell | $ | 168,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 19,999 | $ | 297,999 | $ | 152,500 | $ | 110,000 | $ | 0 | $ | 0 | $ | 19,999 | $ | 282,499 | ||||||||||||||||||
J. M. Cornelius | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | ||||||||||||||||||||||||||||||
J. M. Cornelius(6) | $ | 181,667 | $ | 207,500 | $ | 0 | $ | 0 | $ | 0 | $ | 389,161 | ||||||||||||||||||||||||||||||
L. J. Freeh | $ | 124,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 10,000 | $ | 244,000 | $ | 125,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 0 | $ | 235,000 | ||||||||||||||||||
L. H. Glimcher, M.D. | $ | 142,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 2,000 | $ | 254,000 | $ | 132,500 | $ | 110,000 | $ | 0 | $ | 0 | $ | 2,000 | $ | 244,500 | ||||||||||||||||||
M. Grobstein | $ | 148,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 20,000 | $ | 278,000 | $ | 125,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 20,000 | $ | 255,000 | ||||||||||||||||||
L. Johansson | $ | 114,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 0 | $ | 224,000 | $ | 115,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 0 | $ | 225,000 | ||||||||||||||||||
A. J. Lacy | $ | 106,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 20,000 | $ | 236,000 | $ | 107,500 | $ | 110,000 | $ | 0 | $ | 0 | $ | 20,000 | $ | 237,500 | ||||||||||||||||||
V. L. Sato, Ph.D. | $ | 112,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 0 | $ | 222,000 | $ | 115,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 20,000 | $ | 245,000 | ||||||||||||||||||
T. D. West, Jr. | $ | 106,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 0 | $ | 216,000 | $ | 107,500 | $ | 110,000 | $ | 0 | $ | 0 | $ | 0 | $ | 217,500 | ||||||||||||||||||
R. S. Williams, M.D. | $ | 104,000 | $ | 110,000 | $ | 0 | $ | 0 | $ | 10,500 | $ | 224,500 | $ | 107,500 | $ | 110,000 | $ | 0 | $ | 0 | $ | 12,500 | $ | 230,000 | ||||||||||||||||||
| (1) | Includes the annual retainer (pro-rated for partial-year service), committee chair retainers |
Name | Dollar Amount Deferred | Percentage Allocated to Deferred Share Units | Number of Deferred Share Units Acquired | Dollar Amount Deferred | Percentage Allocated to Deferred Share Units | Number of Deferred Share Units Acquired | ||||||||||||||
L. Andreotti | $ | 0 | — | — | ||||||||||||||||
L. B. Campbell | $ | 0 | — | — | $ | 0 | — | — | ||||||||||||
J. M. Cornelius | $ | 0 | — | — | $ | 0 | — | — | ||||||||||||
L. J. Freeh | $ | 0 | — | — | $ | 0 | — | — | ||||||||||||
L. H. Glimcher, M.D. | $ | 0 | — | — | $ | 0 | — | — | ||||||||||||
M. Grobstein | $ | 37,000 | 100 | % | 1,652 | $ | 31,250 | 100 | % | 1,188 | ||||||||||
L. Johansson | $ | 0 | — | — | $ | 0 | — | — | ||||||||||||
A. J. Lacy | $ | 0 | — | — | $ | 107,500 | 100 | % | 4,171 | |||||||||||
V. L. Sato, Ph.D. | $ | 0 | — | — | $ | 0 | — | — | ||||||||||||
T. D. West, Jr. | $ | 26,500 | 50 | % | 587 | $ | 2,240 | 100 | % | 84 | ||||||||||
R. S. Williams, M.D. | $ | 52,000 | 100 | % | 2,313 | $ | 53,750 | 100 | % | 2,043 | ||||||||||
| (2) | On February |
Name L. B. Campbell J. M. Cornelius L. J. Freeh L. H. Glimcher, M.D. M. Grobstein L. Johansson A. J. Lacy V. L. Sato, Ph.D. T. D. West, Jr. R. S. Williams, M.D. As of December 31, 2010 # of Deferred
Share Units Market
Value 15,657 $ 415,233 8,521 $ 225,974 24,031 $ 637,292 53,582 $ 1,420,983 25,313 $ 671,309 35,813 $ 949,761 20,228 $ 536,446 22,068 $ 585,245 17,229 $ 456,920 31,004 $ 822,221
In addition, Mr. Cornelius received shares of company stock totaling $125,000 in value, which represented the grant date fair value. Such shares were provided pursuant to his Non-Executive Chairman and Transitional Non-Executive Chairman Retainers.
| As of December 31, 2009 | |||||
Name | # of Deferred Share Units | Market Value | |||
L. Andreotti | 0 | $ | 0 | ||
L. B. Campbell | 10,465 | $ | 266,640 | ||
J. M. Cornelius | 4,936 | $ | 125,768 | ||
L. J. Freeh | 18,431 | $ | 469,627 | ||
L. H. Glimcher, M.D. | 46,547 | $ | 1,186,009 | ||
M. Grobstein | 18,500 | $ | 471,387 | ||
L. Johansson | 29,641 | $ | 755,258 | ||
A. J. Lacy | 10,783 | $ | 274,750 | ||
V. L. Sato, Ph.D. | 16,564 | $ | 422,050 | ||
T. D. West, Jr. | 11,877 | $ | 302,625 | ||
R. S. Williams, M.D. | 23,085 | $ | 588,217 | ||
| (3) | There were no option awards granted in |
| As of December 31, 2009 | As of December 31, 2010 | ||||||||||||
Name | # of Stock Options | Intrinsic Value | # of Stock Options | Intrinsic Value | |||||||||
L. Andreotti | 0 | $ | 0 | ||||||||||
L. B. Campbell | 20,810 | $ | 1,300 | 18,181 | $ | 10,160 | |||||||
J. M. Cornelius | 5,000 | $ | 1,300 | 5,000 | $ | 4,575 | |||||||
L. J. Freeh | 2,500 | $ | 1,300 | 2,500 | $ | 3,900 | |||||||
L. H. Glimcher, M.D. | 20,810 | $ | 1,300 | 18,181 | $ | 10,160 | |||||||
M. Grobstein | 0 | $ | 0 | 0 | $ | 0 | |||||||
L. Johansson | 20,810 | $ | 1,300 | 18,181 | $ | 10,160 | |||||||
A. J. Lacy | 0 | $ | 0 | 0 | $ | 0 | |||||||
V. L. Sato, Ph.D. | 0 | $ | 0 | 0 | $ | 0 | |||||||
T. D. West, Jr. | 0 | $ | 0 | 0 | $ | 0 | |||||||
R. S. Williams, M.D. | 0 | $ | 0 | 0 | $ | 0 | |||||||
| (4) | The defined benefit pension plan for directors was discontinued in 1996 and benefits were frozen at that time. None of our current directors participated in this plan. |
| (5) | Amounts |
| (6) | Commencing May 4, 2010, Mr. |
COMPENSATION DISCUSSION AND ANALYSIS
INTRODUCTION
This Compensation Discussion and Analysis (CD&A) aimsis intended to explain to our stockholders how our compensation program is designed and how it operates with respect to our Named Executive Officers (our current CEO, our former CEO, CFO who resigned from the company effective December 31, 2009, and the three other most highly paid executives of our company). James M. Cornelius, our former Chief Executive Officer and current Chairman of the Board of Directors, retired as CEO effective May 4, 2010. This CD&A includes Mr. Cornelius’ compensation information during the time he served as our CEO.
Our CD&A first describes our executive compensation philosophy and how we design our compensation program, with a discussion focusing on the main components of our compensation program. We then detail the process for, and analyze the determinations madedetermination by the Compensation and Management Development Committee (the “Committee”)(Committee) of, the resultant compensation of our Named Executive Officers. Finally, we discuss our 20102011 compensation and benefit program changes, outline other benefits we provide to our Named Executive Officers and describe several of our key executive compensation policies.
EXECUTIVE SUMMARY
In 2009, we successfully completed our transformation to a next-generation biopharmaceutical company facilitated by three significant accomplishments:
2010 Executive Compensation Highlights
the acquisition of Medarex, Inc.;
the split-off of Mead Johnson Nutrition Company; and
| • | Our 3-year and 5-year total stockholder return exceeded that of our peer group average and the |
| • | Our annual and long-term incentives are 100% performance-based |
| • | A significant portion of an executive’s compensation is at risk and tied to |
| • | We eliminated tax gross-ups in |
| • | We have robust share ownership and share retention guidelines and prohibit speculative and hedging transactions |
| • | Our program appropriately mitigates risks |
| • | We have clawback provisions for our incentive awards |
As described in more detail below, in 2009, our Named Executive Officers were compensated based on the successful implementation of our strategy, and the strongsolid financial performance of the company.BMS and their individual performance. In addition, our compensation program was aligned with our goals of strengthening our pay-for-performance philosophy, reducing costs of the compensation program and delivering stockholder value.
20092010 Financial Performance
Our executive compensation program (and thereforeand the compensation of our Named Executive Officers) isOfficers are directly tied to the financial performance of our company. The successful implementation of our biopharmaceutical strategy, the continuing effectssignificant increase in sales of many of our Productivity Transformation Initiative related tokey products, the significant advances in our transformation into a next-generation biopharmaceutical companypipeline, our success in implementing our “string of pearls” strategy including the acquisition of ZymoGenetics, Inc. and the improvement of our net cash position through the divestiture of our last non-core business unit, Mead Johnson,continued expense management all contributed to the positivesolid financial performance of BMS in 2010. The financial measures used in our company ascompensation arrangements in 2010 were applicable to the determination of incentive awards for all eligible employees, including our Named Executive Officers. These financial measures are detailed below:
Financial Measure | Target | Actual | Achievement Level | Target | Actual(2) | Percent of Target | ||||||||
Adjusted Non-GAAP Diluted Earnings Per Share | $1.70 | $1.89 | 111.2% | $2.17 | $2.22 | 102.3% | ||||||||
Net Sales, Net of Foreign Exchange ($=MM)(1) | $19,199 | $19,537 | 101.8% | |||||||||||
| Working Capital plus Capital Expenditures as a % of Net Sales, Net of Foreign Exchange(2) | 18.1% | 15.9% | 112.2% | |||||||||||
Adjusted Net Sales Growth, Net of Foreign Exchange | 6.1% | 4.1% | 67.2 | % | ||||||||||
| Adjusted Working Capital plus Capital Expenditures as a % of Net Sales, Net of Foreign Exchange | 12.5% | 9.6% | 123.2% | |||||||||||
Adjusted Non-GAAP Pretax Profit Margin | 21.9% | 24.5% | 111.9% | 23.5% | 24.4% | 103.8% | ||||||||
| (1) |
| (2) |
Explanation of Financial Measures
TwoEach financial measuresmeasure used to compensate our employees Adjusted Non-GAAP Diluted Earnings Per Share and Adjusted Non-GAAP Pretax Profit Margin, werewas adjusted to exclude operating expenses relatedrelating to (i) the acquisition of Medarex,ZymoGenetics, Inc. and (ii) the extensionportion of our agreement with Otsukathe expense related to U.S. health care reform that permits us to continue marketing Abilify® inwas not quantifiable at the U.S. through April of 2015. These transactions are projected to be accretive intime targets were approved by the long-term, but they required unbudgeted spending in 2009.TheCommittee. The Committee determined after the acquisition of ZymoGenetics, Inc. that it was appropriate to exclude these expenses for compensation purposesthe expense relating to the acquisition of ZymoGenetics, which were not quantifiable when targets were set, so as not to penalize employees in the short-termshort term for making decisions that are projected to create long-term value for our company and itsour stockholders. The amountCommittee also determined after the enactment of adjustment granted equaled the amounthealth care reform legislation that it was appropriate to exclude the portion of unbudgeted spendingthe impact of health care reform that was incurred: $0.04 onnot quantifiable when the targets were approved by the Committee as the employees were not responsible for such impact. The impact of health care reform is a decrease in net sales of $155 million and a decrease in non-GAAP diluted earnings per share basisof $0.055. The impact of ZymoGenetics is an increase in net sales of $15 million and $110 million on a pre-tax basis. decrease in non-GAAP diluted earnings per share of $0.01.
The adjustment appliesadjustments apply to the payoutperformance results under our 2009the 2010 annual bonus plan and the 20092010 portion of our outstanding long-term performance awards, the latter of which are tied to annual financial targets established each year during the respective three-year performance period of each award. The table below displays our year-end results before and after the adjustment:
Financial Measure | Actual | After Adjustment | As Reported | Impact of Health Care Reform | Impact of Zymo-Genetics | After Adjustments | ||||||||||||||
Non-GAAP Diluted Earnings Per Share | $1.85(1) | $1.89 | $ | 2.16 | $ | .055 | $ | .01 | $ | 2.22 | ||||||||||
Non-GAAP Pretax Profit Margin | 23.9%(2) | 24.5% | ||||||||||||||||||
Net Sales Growth, Net of Foreign Exchange(2) | 3.3 | % | 0.8 | % | (0.1 | )% | 4.1 | % | ||||||||||||
Working Capital plus Capital Expenditures as a % of Net Sales, Net of Foreign Exchange(3) | 9.4 | % | 0.2 | % | 0 | % | 9.6 | % | ||||||||||||
Non-GAAP Pretax Profit Margin(4) | 23.8 | % | 0.4 | % | 0.2 | % | 24.4 | % | ||||||||||||
| (1) | As disclosed in our Annual Report on Form 10-K for the fiscal year ended December 31, |
| (2) | Our GAAP |
| (3) | Our working capital plus capital expenditures as a % of |
| (4) | Our GAAP pretax profit margin for 2010 was 31.2%. Non-GAAP pretax profit margin for 2010 was 23.8%. For a reconciliation of GAAP to non-GAAP, see Exhibit 99.2 to the Form 8-K filed on January 27, 2011. |
Total Stockholder Return
In addition to strong financial performance, our total stockholder return for the 3 and 5-year periods ending in 2009 exceeded our peers and the S&P 500 Index. The 5-year stock performance graph is included in the Annual Report accompanying this Proxy Statement. Our compensation program is designed to have a significant portion of compensation paid in the form of equity to tie executives’ interests to the interests of our stockholders. As shown below, our total stockholder return (stock price appreciation plus dividends (TSR)) for the three and five-year periods ending in 2010 exceeded that of our peers (includes companies in our primary and secondary peer groups) and the S&P 500 Index. In 2010, for the second year in a row, we increased our dividends.
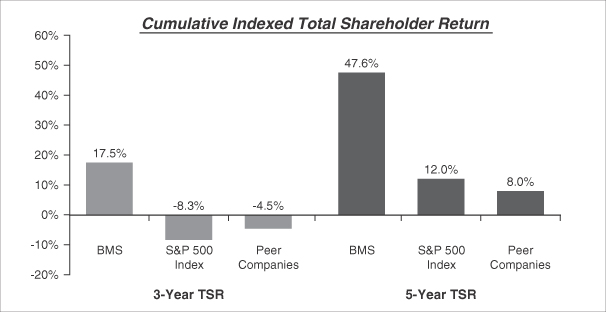
Committee’s Exercise of Negative Discretion to Reduce Incentive Payments
As more fully explained on page 33, the Committee determined that it was appropriate to reduce incentive payouts to levels that reflected the inclusion of the financial performance of the Mead Johnson business unit as Mead Johnson had been a part of the company for nearly the entire year.
20092010 Changes to our Compensation Program
We implemented the following changes to our compensation program in 20092010 to help drivecontinue driving the successful execution of our biopharmaceutical business strategy:
| • |
|
| • | New Long-Term Incentive |
we added total company net sales growth as a performance measure to our 2009 annual incentive program;
we added total company cash flow improvement as a performance measure to our 2009-2011 long-term performance award; and
we changed our cash flow measure from a dollar target to an improvement ratio (working capital plus capital expenditures as a percent of net sales).
Both our annual and long-term programs now have the same performance measures and weightings: U.S. non-GAAP earnings per share (weighted 50%), net sales (weighted 25%) and working capital plus capital expenditures as a percent of net sales (weighted 25%). This approach focused the organization on balancing top-line growth with strategic bottom-line growth and the efficient use of cash. In addition, we established that sales achievement would be measured excluding the effect of foreign exchange. Measuring sales achievement net of foreign exchange eliminates a factor which management does not control. We believe the diversity of metrics used in our incentive programs, in conjunction with the long-term vesting of equity awards and the use of share ownership and retention guidelines, are appropriate and do not encourage excessive risk-taking behaviors.
| • |
|
| • | Market Share Units: Market share units comprise 40% of the new long-term |
2010 MANAGEMENT CHANGES
On January 1, 2010, the Committee appointed Mr. Bancroft to serve as our Acting Chief Financial Officer and on April 8, 2010, Mr. Bancroft became our Chief Financial Officer. On May 4, 2010, Mr. Cornelius retired as Chief Executive Officer of our company and Mr. Andreotti became our Chief Executive Officer. Messrs. Andreotti and Bancroft are subject to the same policies and programs that are applicable to all other Named Executive Officers.
Upon retirement as CEO, Mr. Cornelius was entitled to receive certain benefits pursuant to our standard retirement policy. Specifically, Mr. Cornelius was eligible to:
receive a pro rata bonus for 2010 based on his service and performance through May 4, 2010.
have the full remaining exercise term to exercise his outstanding stock options.
receive pro rata payouts under the performance share unit awards that were outstanding as of his retirement date, based on the period of his employment.
EXECUTIVE COMPENSATION PHILOSOPHY
Our executive compensation philosophy is based on two core elements: to pay for performance and to provide a competitive compensation package. Each of these elements is described below:
Pay for Performance: We structure our compensation program to align the interests of our senior executives with the interests of our stockholders. We believe that an employee’s compensation should be tied directly to helping us achieve our mission and deliver value to our stockholders. Therefore, a significant part of each executive’s pay depends on his or her individual performance against financial and operational objectives as well as meeting key behavioral standards. We also believe that a significant amount of compensation should be at risk. A substantial portion of an executive’s compensation, therefore, is in the form of equity awards that tie the executive’s compensation directly to creating stockholder value and achieving financial and operational results.
Competitive Pay: We believe that a competitive compensation program is an important tool to help attract and retain talented employees capable of leading our business in the highly complex and competitive business environment in which we operate. We aimintend to pay our executives at approximately the median level of pay of our peer group when targeted levels of performance are achieved. In certain circumstances, we may target pay above or below the competitive median to help attract or retain executives, as necessary, or to recognize differences in their qualifications, responsibilities, role criticality and/or potential. By providing compensation that is competitive with our peer companies, we reduce the risk that our executivescompetitors can be recruited away.successfully recruit our executives.
In addition, our compensation program is designed with the following principles in mind:
to pay our employees equitably relative to one another based on the work they do, the capabilities and experience they possess, and the performance they demonstrate;
to promote a non-discriminatory work environment that enables us to benefit from the diversity of thought that comes with a diverse workforce;
to motivate our executives to deliver high performance with the highest integrity; and
to continue to focus on good corporate governance practices by implementing compensation best practices and corporate policies, several of which are described in greater detail beginning on page 43.44.
We believeBased on our review of our executive compensation arrangements as detailed on page 11, our Committee believes that our compensation program is designed so asdoes not to encourage executives to take unreasonable risks that may harm stockholder value. We have achievedOur compensation program achieves this by striking an appropriate balance between short-term and long-term incentives, by using a diversity of metrics to assess performance under our incentive programs, and by using different forms of long-term incentives, by placing caps on our incentive award payout opportunities, following equity grant practices that limit potential for timing awards and by having stock ownership and stock retention requirements.
OUR COMPENSATION PROGRAM DESIGN
This section will explain how we determine the design of our executive compensation program.
Compensation and Management Development Committee
The Compensation and Management Development Committee (“Committee”) is responsible for providing oversight of our executive compensation program for the Named Executive Officers as well as other members of senior management. The Committee is responsible for recommendingsetting the compensation of the Chief Executive Officer and approving the compensation of all of the other Named Executive Officers.
The Committee annually reviews and evaluates the executive compensation program to ensure that the program is aligned with our compensation philosophy. The “Committees of ourOur Board” section on page 8 discusses the duties and responsibilities of the Committee in more detail.
Independent Compensation Consultant
In September 2009, the Committee retained Compensation Advisory Partners, LLC (CAP) as its independent compensation consultant to provide executive compensation services to the Committee. The Committee decided to retain CAP as its independent compensation consultant when the individual consultants that the Committee had worked with since 2002 left Mercer (US) Inc., the Committee’s former consultant, to form CAP.CAP is not affiliated with Mercer. CAP reports directly to the Committee, and the Committee directly oversees the fees paid for services provided by CAP. The Committee instructs CAP to give advice to the Committee independent of management and to provide such advice for the benefit of our company and stockholders. CAP does not provide any consulting services to the companyBMS beyond its role as consultant to the Committee.
Prior to September 2009, Mercer (US) Inc. served asIn 2010, CAP provided the Committee’s compensation consultant. While the members of the Mercer consulting team giving advice to the Committee could not provide any other service to the company, our company business units were able to retain other Mercer consultants for discrete human resource services in the U.S. and abroad. Our business units were required to engage in a competitive bidding process when selecting a company to provide human resource services and Mercer was able to submit a bid along with other viable competitors. The Committee reviewed the overall fees incurred by the Committee and the fees incurred by company business units for services provided by Mercer. While the Committee did not pre-approve the non-executive compensation services, it reviewed Mercer’s internal guidelines and practices to guard against conflicts and to ensure the objectivity of their advice. The Committee believes that the services Mercer provided to company business units did not impact the advice Mercer provided to the Committee on executive compensation matters.
During the time Mercer served as the Committee’s consultant in 2009, the fees paid to Mercer were as follows:
Executive Compensation Services(1) | $ | 235,311 | |
Non-Executive Compensation Services(2) | $ | 1,537,784 |
In 2009, the compensation consultants retained by the Committee:following services:
participatedParticipated in the design and development of our executive compensation program;
providedProvided competitive benchmarking and market data analysis;
providedProvided analysis and information about industry trends relating to the compensation of our CEO and Named Executive Officers;
providedProvided an annual review of our change-in-control benefits, including analyzing these benefits againstas compared to those provided by our peer companies and best practices;
reviewedReviewed and advised on all materials provided to the Committee for discussion and approval; and
attendedAttended all of the Committee’s regularregularly-scheduled meetings in 20092010 at the request of the Committee.
Role of Company Management
The CEO makes recommendations to the Committee concerning the compensation of the other Named Executive Officers and other senior management. In addition, the CEO and CFO are involved in settingrecommending for the Committee’s approval the business goals that are used as the performance
goals for the annual and long-term incentive plans, subject to Committee approval.plans. The Senior Vice President of Human Resources, Public Affairs and Philanthropy works closely with the Committee, the Committee’s independent compensation consultant and management to (i) ensure that the Committee is provided with the appropriate information to make its decisions, (ii) propose recommendations for Committee consideration, and (iii) communicate those decisions to management for implementation.
Peer Group Analysis
Our executive compensation program seeks to provide total compensation, when targeted levels of performance are achieved, at approximately the median of the pay levels provided by a designated peer group of U.S. companies. We define total compensation as base salary plus annual target bonus plus the target value of long-term incentives. We use competitiveevaluate median pay levels of our peers to maintain our compensation program guidelines on an annual basis (i.e., our salary structure, the leveling of our positions, our annual target bonus levels, and our long-term incentive award guidelines). We also use competitive pay levels of our peers to help in determining individual pay decisions (i.e., base salary levels, the size of salary adjustments, and the size of long-term incentive awards). Paying at the competitive median of our peers when targeted levels of performance are achieved allows us to attract and retain the talent we need to run theour business while also enabling us to maintain a competitive cost base with respect to compensation expense.
Peer Group
Our primary peer group in 20092010 consisted of the following companies:
Abbott Laboratories | Gilead Sciences Inc. | |
Amgen Inc. | Johnson & Johnson | |
Biogen Idec Inc. | Merck & Co. | |
Eli Lilly and Company | Pfizer, Inc. | |
Genzyme Corporation | ||
In 2010, we added three companies, Biogen Idec Inc., Genzyme Corporation and Gilead Sciences Inc., to our primary peer group in light of consolidation in the industry that occurred in 2009. We believe this peer group was appropriate given the unique nature of the pharmaceutical/biotechnology industry. We note that theThe companies in our primary peer group varyvaried in size, but we do notsize. BMS approximated the median in both revenue and market capitalization amongst our primary peer group. We believe that company size, however, should not be the only factor in determining a peer group. Instead, we believe emphasis should be placed on whether a company competes directly with us for unique pharmaceutical/biotechnology talent. The companies in our 20092010 primary peer group represented our primary competitors for executive talent and operated in a similarly complex regulatory and research-driven environment. We also added a secondary peer group comprised of the nine companies in our primary peer group plus five companies based outside the U.S. The five foreign companies included in our secondary peer group are: AstraZeneca PLC, GlaxoSmithKline PLC, Roche Holding Ltd., Novartis AG and Sanofi-Aventis SA. This secondary peer group serves as an extra point of reference for the Committee given the global nature of our business and the fact that we compete for talent on a global basis. We monitor the composition of our peer groupgroups regularly and make changes when appropriate. For 2010, we will change our peer group in light of consolidations in the industry that occurred in 2009.
The Committee’s independent compensation consultant annually conducts a review of the compensation for our Named Executive Officers using compensation information compiled from the proxy statement disclosures ofmade by our primary peer group. While we target total compensation at median, the Committee took multiple factors into consideration when setting the target pay package for each Named Executive Officer in 2010, including the length of time in the officer’s current role, individual roles and responsibilities and relative company size. The following table shows the competitive pay position of our Named Executive Officers in 2009:2010:
Executive | Executive’s Total Target Compensation versus Market Pay Percentile(1) | |
|
| |
|
| |
Lamberto Andreotti |
| |
Charles A. Bancroft | Between 25th and 50th Percentiles | |
Elliott Sigal, M.D., Ph.D. |
| |
Anthony C. Hooper |
| |
Sandra Leung | Between 25th and 50th Percentiles | |
James M. Cornelius | Not Applicable(2) |
| (1) | Percentile figures reflect |
| (2) |
|
DETERMINING THE INDIVIDUAL COMPENSATION OF OUR EXECUTIVES
This section will explain how the 20092010 compensation was determined for each of our Named Executive Officers, including our former CFOCEO who resignedretired effective December 31, 2009.May 4, 2010.
Our executive compensation program is designed to provide value to the executive based on the extent to which (i) individual performance, (ii) company performance versus annual budgeted financial targets and (iii) total return to stockholders (stock price appreciation plus dividends) meet, exceed or fall short of expectations. We believe this approach, with a significant emphasis on long-term compensation, serves to focus the efforts of our executives on the attainment of sustained long-term growth and profitability for the benefit of our company and our long-term stockholders.stockholders while demonstrating high ethical standards.
When determining individual award levels, the Committee gives equal weight to (i) individual performance against financial and operational objectives that are linked to our business strategy and total stockholder return (“Results”) and (ii) an executive’s demonstration of the values and behaviors defined in the Bristol-Myers Squibb Commitment and Core BMS Behaviors (“Behaviors”) | 2010 Core BMS Behaviors
Leads Strategically
|
Performance Management System
Our performance management system involves an annual review of all executives, including the Named Executive Officers, which measures individual performance over the course of the previous year. The system assists in ensuring that each executive’s compensation is tied to the financial and operational performance of theour company, to stockholder return, and to the executive’s demonstration of the Core BMS Behaviors and the values embodied in the BMS Commitment. The Committee conducts the assessment process for our CEO. The CEO conducts the assessment for all of our other Named Executive Officers. The assessment for each Named Executive Officer is then reviewed and approved by the Committee.
Each executive wasis assessed on both “Results” and “Behaviors”. The Committee conducted the assessment process for our CEO. The CEO conducted the assessment for Mr. Andreotti and Dr. Sigal. Mr. Hooper’s assessment was conducted by his first-level manager (Mr. Andreotti) and reviewed by the CEO. Mr. Huet, our former CFO, was not assessed due to his resignation from our company. The assessment for Messrs. Andreotti and Hooper and Dr. Sigal was then reviewed and approved by the Committee.
The Committee useduses these assessments as the basis for making individual compensation decisions.1 The assessments described below pertain to 20092010 performance and were used to help the Committee determine the size of each Named Executive Officer’s 20092010 annual bonus payment. Prior-year assessments, as disclosed in our 20092010 proxy statement, were used by the Committee to determine the size of each Named Executive Officer’s 2009 stock option and restricted stock unitthe 2010 long-term incentive awards which were granted in March of 2009. Prior-year assessments were also used2010 to determine eligibility for the flat 2% merit increase as described earlier, which were effective in April of 2009.each Named Executive Officer, except Mr. Cornelius.
Individual Performance
When determining the individual compensation decisions,2010 annual incentive payments and the 2011 long-term incentive awards, the Committee considered the generally strong financial performance of theour company against preset targets as set forthfinancial measures identified on page25. In addition to the company’s financial performance, the Committee considered individual performance factors when determining 2009 annual incentive payments (as well as 2010 equity awards), including 27 and general operational goals, such as increasing sales of our products, which had a direct impact on the preset financial goals.
For Mr. Cornelius,measures. In addition, the Committee consideredplaced an emphasis on the successful transformation to a pure biopharmaceutical company throughrequirement that the partial initial public offering and subsequent split-off of the Mead Johnson business unit and the successful implementation of the “String of Pearls” strategy through the acquisition of Medarex, Inc. and the licensing deals with Teijin Pharma Limited, ZymoGenetics and Alder Biopharmaceuticals. The Committee also considered his demonstrated commitment to the highest standards of transparency, ethics and integrity.executives achieve their goals while demonstrating high ethical standards.
For Mr. Andreotti, the Committee considered the increase in worldwide sales of several key products, his leadership in taking steps to mitigatein: (i) delivering the solid 2010 financial results shown on page 27, while effectively managing the impact of U.S. health care reform and the upcoming lossglobal economic crisis; (ii) effectively developing and preparing for the launch of exclusivity for Plavix®several new products; (iii) streamlining and simplifying our organizational structure and business processes; and (iv) creating long-term sustainability through an industry-leading R&D pipeline as well as industry-leading expense management. The Committee also considered his agility at assuming the CEO role, creating and energizing his Senior Management Team, and mobilizing the organization behind our business strategy.
For Mr. Bancroft, the Committee considered his key role in the U.S.,achievement of our financial results in 2010. The Committee also considered his strong leadership over strategic initiatives and our capital allocation strategy aimed at creating longer-term sustainability, including financial oversight of the “string of pearls” strategy, continuous productivity efforts that are projected to yield an additional $1.2 billion in driving broad cultural and operational changes consistent with our biopharma strategy, particularly in the areas of talent management, decision-makingcost savings by 2013, and the furthering of continued efficienciesinitiative to retire $750 million in the company’s manufacturing network.debt.
For Dr. Sigal, the Committee considered his performance in exceedingleading an R&D organization recognized externally as one of the company’s goals for sustainingmost innovative, productive and successful in the industry. In 2010, the R&D pipeline byorganization’s accomplishments included: (i) advancing 4 productssubmitting to full developmentthe FDA six major New Molecular Entity filings versus a target of 3,five; (ii) receiving eight significant regulatory approvals meeting a target of eight; (iii) achieving proof of confidence for six compounds versus a target of three; and (iv) producing 14fifteen Early Candidate Nominations versus a target of 13 and (iii) achieving 15 first-in-human submissions versus a target of 8. In addition, the Committee considered his strong leadership of the R&D organization to deliver a high level of productivity while building one of the strongest near-term pipelines in the industry.fourteen.
For Mr. Hooper, the Committee considered the increase in U.S. sales of severalmost key products.products and strong profitability in the Intercontinental and Japan/Pacific/Asia regions. The Committee also considered his strongleadership in: (i) planning for several critical product launches during the fourth quarter 2010 through the first quarter 2011; (ii) driving innovative projects globally regarding access and customer support to gain market share and increase competitiveness; and (iii) achieving best-in-class expense control in the U.S. commercial business.
For Ms. Leung, the Committee considered her performance in providing consistently sound legal advice to senior management and the Board of Directors. The Committee also considered her leadership in drivingtransforming the Law Department to support our biopharmaceutical strategy, which included: (i) improving the quality of legal support while reducing expenses; (ii) streamlining and simplifying business processes; and (iii) accelerating culture change and energizing employeesdriving innovation to improve business results.
For Mr. Cornelius, the Committee considered his leadership in successfully transforming Bristol-Myers Squibb into a biopharmaceutical company positioned to deliver strong performance in an increasingly complexshort-term results and competitive environment.sustainability over the long term through its unique strategy and strong R&D pipeline and the successful transition of CEO responsibilities to Mr. Andreotti.
THE COMPONENTS OF OUR 20092010 COMPENSATION PROGRAM
The main components of our executive compensation program in 20092010 were:
Base Salary
Annual Incentive Award
Long-Term Incentives
Long-Term Performance Awards (Performance Share Units)Units (Long-Term Performance Awards)
Stock Options
Restricted StockMarket Share Units
The following chart shows the 20092010 compensation mix for these elements based on targeted compensation for our Named Executive Officers:Officers, excluding Mr. Cornelius:
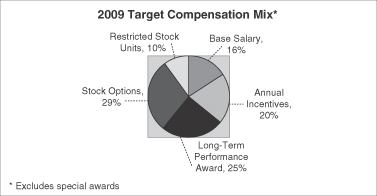
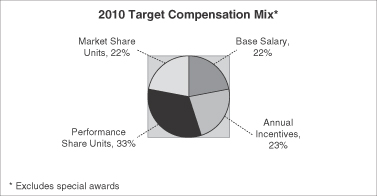
This target mix supports the core elements of our executive compensation philosophy by emphasizing long-term incentives while providing competitive short-term components. Below, we explain how each of these components is set and describe certain changes we made to these components in 2009.2010. The changes noted were implemented to provide for the continued alignment of our compensation program with the core elements of our compensation philosophy. The specific pay decisions with respect to our Named Executive Officers are also detailed.
Base Salary
Base salaries are a tool used both to help keep us competitive and to help us retain talent. The base salaries of our executives are set based primarily upon the pay levels of comparable positions within our primary peer group and the unique qualifications and experience of the individual executives. Merit increases for our executives are determined based upon both the performance of an individual and the size of our merit increase budget in a given year. We review results of surveys that forecast what other companies’ salary increase budgets will be. We typically set our annual salary increase budgets based upon the median of such forecasts. Salary adjustments are also typically granted when executives assume significant increases in responsibility.
In 2009,2010, we scaled back our merit budgets relative to the competitive median as a cost-savings measure. Within the U.S., we granted flat 2%did not grant annual merit increases to employees who were ratedour Named Executive Officers as fully performing or higher on both Results and Behaviors. Employees rated below the fully-performing level typically received no salary increase. Consistent with this policy, an additional cost-savings measure.
Mr. Andreotti Dr. Sigal and Mr. Huet each received a 2%12% salary increase on April 1, 2009, their regularly-scheduled merit review date. Mr. Cornelius elected not to receive a salary increase in 2009.
Mr. Hooper received a 14.3% salary increase on March 3, 2009May 4, 2010 in connection with his promotion to the position of President, Americas. InChief Executive Officer. This salary increase, in combination with the other components of Mr. Andreotti’s new CEO compensation package, positioned his new role, Mr. Hooper assumed general management responsibility for Canada, Latin America,total compensation at approximately the Middle East, Africa and certain distributor markets,25th percentile among CEOs in addition to maintaining his responsibility for U.S. Pharmaceuticals. Mr. Hooper’s new totalour primary peer group.
compensation package was positioned at the competitive median of our peer group for similarly-sized general management positions.
Annual Incentives
Annual incentive awards are designed to reward the Named Executive Officers for achieving short-term financial and operational goals and to reward their individual performance, consistent with our pay-for-performance philosophy. A Named Executive Officer’s annual incentive award opportunity is expressed as a percentage of base salary as determined by the individual’s grade level.
Under the company’s 2009our 2010 bonus plan design, as described earlier, three corporate-wide measures—non-GAAP diluted earnings per share (weighted 50%), non-GAAP net sales growth, net of foreign exchange (weighted 25%) and working capital plus capital expenditures as a percent of net sales net of foreign exchange (weighted 25%)—serve to fund the company’sour bonus pool. Individual target bonuses, in turn, may be increased or decreased based upon theour company’s performance against these corporate-wide measures. Assuming fundingThe table below shows the resulting payout percentage of the financial measures used for our 2010 bonus pool,plan:
Financial Measure | Percent of Target | Resulting Payout Percentage | ||||||
Adjusted Non-GAAP Diluted Earnings Per Share | 102.3 | % | 109.23 | % | ||||
Adjusted Net Sales Growth, Net of Foreign Exchange | 67.2 | % | 91.44 | % | ||||
(Net Sales, Net of Foreign Exchange, as a dollar amount)1 | (98.4 | %) | ||||||
Adjusted Working Capital plus Capital Expenditures as a % of Net Sales, Net of Foreign Exchange | 123.2 | % | 162.79 | % | ||||
Total | 118.17 | % | ||||||
| (1) | We are providing achievement levels of a net sales, net of foreign exchange, dollar financial measure for informational purposes. Our net sales percent growth measure was designed to provide the same level of achievement and results as the net sales dollar measure we had used in prior years. Because net sales dollar achievement is more transparent to employees than net sales percent growth achievement, the Committee has decided to revert to a net sales dollar target as a financial measure for 2011. |
Assuming the achievement of the financial measures, the actual bonus an executive receives is based entirely on individual performance. As described above, individual performance is assessed on the two dimensions of our performance management process—Results and Behaviors. Additionally, the bonuses of our Named Executive Officers and other Senior Management CouncilTeam members may be modified up or down based on the extent to which each executive meets pre-established diversity placement goals and demonstrates other actions that promote diversity (e.g., ensuring that diversity candidates are considered for developmental opportunities; including diversity candidates in succession plans; mentoring employees with diverse backgrounds; leading an employee affinityresource group; and holding staff members accountable for advancing our diversity objectives). We place an emphasis on diversity in our annual incentive program because we believe a diverse workforce, which engenders diversity of thought and perspective, is a source for creating a competitive advantage. In 2010, the Committee determined not to modify any of the Named Executive Officer’s bonuses because the Named Executive Officers demonstrated their commitment to diversity on a relatively equal basis by serving as executive sponsors of several employee resource groups, hosting various diversity awareness and advancement forums and ensuring that our talent acquisition slates included viable diversity candidates.
We believe this approach for determining incentive award payments balances the need to consider overall company performance, results specific to an executive’s functional area of responsibility, and the executive’s ability to achieve results vs. objectives on an individual level while also demonstrating the Core BMS Behaviors. The recommended payments are reviewed and approved by the Committee in the first quarter of the year following the performance year, and the awards are paid by March 15th.
Exercise of Negative Discretion
In 2009, the company exceeded target on all three performance measures. Our annual bonus plan provides that discontinued operations be excluded from financial results for payout purposes. Therefore, the financial measures excluded the performance of the Mead Johnson business unit, which was split-off from the company on December 23, 2009. The Committee, however, determined that because the company managed Mead Johnson for nearly the entire year, it was more appropriate to include the financial performance of Mead Johnson for incentive payout purposes, which would result in lower payouts. The Committee exercised its negative discretion and reduced the annual incentive payouts to levels that reflect the inclusion of the financial performance of the Mead Johnson business unit.
The table below shows the company’s performance versus target, the factor used to adjust target bonuses and the factor used to fund the company bonus pool:
Measure | Target | Actual | % Achieve | Target Bonus Adjustment Factor(1) | Bonus Pool Funding Factor(1) | ||||||||||||
EPS | $ | 1.70 | $ | 1.89 | 111.2 | % | 146.00 | % | 153.50 | % | |||||||
Sales ($=MM) | $ | 19,199 | $ | 19,537 | 101.8 | % | 101.80 | % | 109.30 | % | |||||||
Work Cap + CapEx (%) | 18.1 | % | 15.9 | % | 112.2 | % | 151.00 | % | 158.50 | % | |||||||
Total Before Adjustment | 136.20 | % | 143.70 | % | |||||||||||||
Total After Adjustment | 133.18 | %(2) | 140.68 | %(2) | |||||||||||||
The actual annual incentives paid to our current Named Executive Officers are shown in the table below and also reported in the Summary Compensation Table in the Non-Equity Incentive Plan Compensation column:
Executive | Target Bonus | Adjusted Target Bonus | Actual Payout | % of Target | Target Bonus | Adjusted Target Bonus(1) | Actual Payout(2) | % of Target | |||||||||||||||||||
Mr. Cornelius | $ | 2,250,000 | $ | 2,996,550 | $ | 4,494,825 | 200% | ||||||||||||||||||||
Mr. Andreotti | $ | 1,649,970 | $ | 2,197,430 | $ | 3,076,402 | 187% | $ | 1,962,987 | $ | 2,319,662 | $ | 3,131,544 | 159.5 | % | ||||||||||||
Mr. Bancroft | $ | 662,500 | $ | 782,876 | $ | 978,595 | 147.7 | % | |||||||||||||||||||
Dr. Sigal | $ | 1,218,000 | $ | 1,622,132 | $ | 2,189,879 | 180% | $ | 1,224,000 | $ | 1,446,401 | $ | 1,952,642 | 159.5 | % | ||||||||||||
Mr. Hooper | $ | 759,992 | $ | 1,012,157 | $ | 1,113,373 | 147% | $ | 800,000 | $ | 945,360 | $ | 1,276,236 | 159.5 | % | ||||||||||||
Ms. Leung | $ | 540,000 | $ | 638,118 | $ | 797,648 | 147.7 | % | |||||||||||||||||||
Mr. Cornelius(3) | $ | 937,575 | $ | 1,107,932 | $ | 1,495,709 | 159.5 | % | |||||||||||||||||||
Consistent with
| (1) | Adjusted to reflect financial performance earned at 118.17%. |
| (2) | Adjusted to reflect individual performance. |
| (3) | Mr. Cornelius’ target bonus was prorated to reflect his service through May 4, 2010 as CEO. |
As set forth in the bonus plan formula describedtable above, each Named Executive Officer’s target bonus was earned at 133.18%118.17% based on the company’s strongsolid financial results. Then, an individual performance payout factor was applied to each of the adjusted target bonuses. In determining the individual annual incentives paid to our Named Executive Officers, the Committee considered the 20092010 performance of each Messrs. Cornelius, Andreotti and Hooper and Dr. Sigalexecutive as described under “Individual Performance” on page 31.33. The Committee approved individual performance factors ranging between 110%125% and 150%135% for each of Messrs. Cornelius, Andreotti and Hooper and Dr. SigalNamed Executive Officer based on the strong individual performance results. Having resigned at the end of 2009, Mr. Huet was not eligible to receive an annual bonus for 2009.results described above.
Long-Term IncentivesIncentive Program
Long-term incentives are designed to tie executive interests to the interests of our stockholders. The ultimate value of long-term awards is driven by stock price and dividends, which provide a direct link to the creation of stockholder value. In addition, our long-term incentive program is designed to reward individual performance. In 2009,2010, we offered threetwo long-term award vehicles, each of which served a different purpose:
Long-Term Performance Share Unit Awards (performance share unit awards)(Long-Term Performance Awards) to reward the achievement of internal financial goals; and
Stock OptionMarket Share Unit Awards to reward the creation of incremental stockholder value;value and
Restricted Stock Unit Awards to help us retain key talent.
We believe our long-term incentive program serves the best interests of our stockholders by focusing the efforts of our executives on key financial drivers of long-term success and on total stockholder return while enabling us to reduce expenses. We redesigned our long-term incentive program to achieve the following objectives that support our biopharmaceutical business strategy:
The entire long-term incentive program for executives is performance-based.
The new design applies uniformly to all levels of executives, thus promoting organizational alignment with our biopharmaceutical strategy.
With vesting and payout spread over several years, including the payouts relating to dividend equivalents, the new program promotes greater retention of our executives.
The elimination of stock option awards and the reduction in the target value of long-term incentive awards resulted in the 2010 compensation program being approximately 17% less costly than the 2009 compensation program.
The new program offers the opportunity to earn dividend equivalents on performance share units and market share units only when the underlying award is earned.
The following graph shows the 20092010 target mix of our long-term incentives for allour Named Executive Officers:Officers, excluding Mr. Cornelius who did not receive 2010 awards:
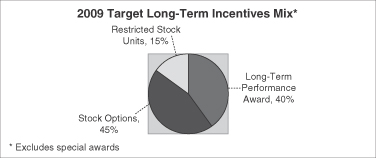
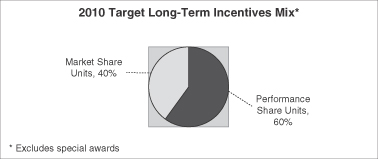
Annual Equity Award Grants
Annual equity award grants are typically made on the first Tuesday in March to coincide with meetings of the Committee and the full Board of Directors. We believe that consistent timing of equity award grants is a good corporate governance practice that reduces the risk of selecting a grant date with a preferential stock price.
AsUnder our long-term incentive program, the chart below details, the useCommittee establishes target awards, expressed in dollars, at each grade level. The value of performance-based exercise thresholds for our stock options, combinedthese target awards, taken in conjunction with the increased emphasis placed onother components of our Long-Term Performance Award Program in recent years, ensured that approximately 85%pay program, enables us to achieve our overall target level of competitive compensation.
Based upon individual performance, an executive may receive a long-term incentive award ranging from 0% to 150% of the target award. Once the grant value is established for each executive, 60% of the value is converted into performance share units and 40% into market share units.
In March of 2010, each Named Executive Officer, excluding Mr. Cornelius, received new long-term incentives mix for allincentive awards. In determining the size of individual awards granted to our Named Executive Officers, including the Committee considered the prior-year performance of each executive. The Committee approved individual awards ranging from 120% to 140% of the target value for all of the Named Executive Officers, except for Mr. Bancroft, based on strong individual performance results described under “Individual Performance” on page 33. The Committee determined it was appropriate for Mr. Bancroft to receive individual awards at 100% of the target value, despite his strong individual performance, because he had just started his new role as Acting Chief Financial Officer in January 2010.
Effective May 4, 2010, upon becoming our CEO, was tiedMr. Andreotti received additional long-term incentive awards valued at $3,475,001, which brought his total value for 2010 to specific$7,500,000, the long-term incentive target for his new position approved by the Committee. Consistent with our long-term incentive program, Mr. Andreotti received 60% of the May grant as performance criteria, thus supporting our pay-for-performance philosophy.
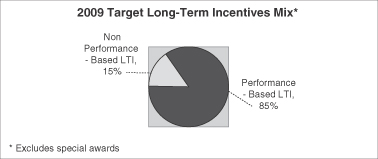
Long-Term Performance Results Under Our Outstanding Performance Share Unit Awards: This type of award, which is expressed in the form
The payout of performance share units,unit awards is based on three-year performance cycles. The target number of units in an executive’s award is tied to his or her grade level. The higher an executive’s grade level, the larger the target award because executives in successively higher grade levels have correspondingly greater levels of responsibility.
In 2007, the Committee modified the design of the long-term performance award program. Starting with the 2007-2009 award, the Committee now approves annual performance targets at the beginning of each year of the three-year performance cycle. Under the prior design, three-year cumulative targets were set at the beginning of each cycle. The timing of payouts remains unchanged under the new design—cycle, and awards continue to beare payable in the year following the end of the three-year cycle. The newThis design provides for a better link between performance and payout because it eliminates the need to project performance beyond one year. This closer line of sight will help to avoid situations where
unforeseen events lead to performance targets that are either overstated or understated and do not appropriately support the company’sour pay-for-performance philosophy. We also eliminated a feature used in previous years in which payouts were adjusted based upon our three-year total stockholder return versus that of our competitors because, under Internal Revenue Code Section 162(m) rules governing performance- based compensation, it was impractical to combine a three-year total stockholder return measure with performance targets that are set annually. Given that long-term performancethese awards are denominated and paid in shares, however,there is a strong link to stockholder return remains.return.
The Committee established a 2010 non-GAAP pretax earnings goal of $3,748 million for the purpose of preserving tax deductibility of 2010 payouts under this award pursuant to IRC Section 162(m). The company’s actual non-GAAP pretax earnings for 2010 of $4,642 million exceeded the established goal, thus enabling (i) payouts under the 2008-2010 regular and special long-term performance awards and (ii) banking of shares under the 2009-2011 and 2010-2012 long-term performance awards
2007-20092008-2010 Award: The 2007-20092008-2010 award is based 50% on non-GAAP diluted earnings per share and 50% on non-GAAP net sales. As described earlier, the company began using a net sales, performance measure in 2009 that is net of foreign exchange. AnIn 2008 and 2009, net sales were measured in dollar terms; in 2010, a percent growth metric was used. Under the 2010 earnings per share metric, achievement of 92% on a given performance measure would yield80% of target yields a payout of 45%32.5% of target for that portion of the award. Achievementaward; achievement below 92% would80% of target will yield no payout. Achievementpayout; and achievement levels of 110%115.7% or higher would yieldof target yields a maximum payout of 220%167.5% for that portion of the award. One-thirdUnder the 2010 net sales growth metric, 0.1% growth over prior year yields a payout of 52.5% of target for that portion of the award was tied to 2007 targets, one-third to 2008 targets,award; growth below this level yields no payout; and growth of 11.8% or higher will yield a maximum payout of 167.5% for that portion of the final third to 2009 targets. Payout results relating to 2007 and 2008 were banked, but did not vest until the payable date in 2010. The entire award was paid in March of 2010.award.
The 2007-20092008-2010 award is payable at 163.73%116.94% of target award. The following table summarizes the company’s performance and payout results relating to this award:
Year | Measure | Target | Actual | % Achieve | % Payout | Measure | Target | Actual | % of Target | % Payout | |||||||||||||||||||||||
| 2007 | EPS | $ | 1.25 | $ | 1.34 | (1) | 107.2 | % | 178.00 | % | |||||||||||||||||||||||
Sales ($=MM) | $ | 19,262 | $ | 19,977 | 103.7 | % | 127.10 | % | |||||||||||||||||||||||||
Annual Total | 152.60 | % | |||||||||||||||||||||||||||||||
| 2008 | EPS | $ | 1.54 | $ | 1.74 | 113.0 | % | 220.00 | % | EPS | $ | 1.54 | $ | 1.74 | 113.0 | % | 155.00 | % | |||||||||||||||
Sales ($=MM) | $ | 19,627 | $ | 20,597 | 104.9 | % | 144.10 | % | Sales ($=MM) | $ | 19,627 | $ | 20,597 | 104.9 | % | 110.80 | % | ||||||||||||||||
Annual Total | 182.10 | % | Annual Total | 132.90 | % | ||||||||||||||||||||||||||||
| 2009 | EPS | $ | 1.70 | $ | 1.89 | 111.2 | % | 220.00 | % | EPS | $ | 1.70 | $ | 1.89 | 111.2 | % | 146.00 | % | |||||||||||||||
Sales ($=MM) | $ | 19,199 | $ | 19,537 | 101.8 | % | 109.00 | % | Sales ($=MM) | $ | 19,199 | $ | 19,537 | 101.8 | % | 101.80 | % | ||||||||||||||||
Annual Total | 164.50 | % | Annual Total | 123.90 | % | ||||||||||||||||||||||||||||
Adjusted Annual Total(2) | 156.50 | %(2) | Adjusted Annual Total | 117.55 | %(1) | ||||||||||||||||||||||||||||
| 2010 | EPS | $ | 2.17 | $ | 2.22 | (2) | 102.3 | % | 109.92 | % | |||||||||||||||||||||||
Three-Year Total | 163.73 | % | Sales Growth(%) | 6.1 | % | 4.1 | %(2) | 67.2 | % | 90.80 | % | ||||||||||||||||||||||
| Annual Total | 100.36 | % | |||||||||||||||||||||||||||||||
| Three-Year Total | 116.94 | % | |||||||||||||||||||||||||||||||
| (1) |
| Reflects the Committee’s use of negative discretion. Our long-term performance award program provides that discontinued operations be excluded from financial results for payout purposes. Therefore, the financial measures excluded the performance of the Mead Johnson business unit, which was split-off from |
2008-2010 Award: The 2008-2010 award is similar in design to the 2007-2009 award, except that the payout schedule differs. Under the 2008-2010 award, achievement of 80% on a given performance measure will yield a payout of 40% of target for that portion of the award; achievement below 80% will yield no payout; and achievement levels of 115% or higher will yield a maximum payout of 165% for that portion of the award. The award is based 50% on non-GAAP diluted earnings per share and 50% on non-GAAP net sales. As described earlier, the company began using a net sales performance measure in 2009 that is net of foreign exchange.
Below are the company’s performance and banked payout results for the first two years of the 2008-2010 award:
Year | Measure | Target | Actual | % Achieve | % Payout | |||||||||
| 2008 | EPS | $ | 1.54 | $ | 1.74 | 113.0 | % | 155.00 | % | |||||
Sales ($=MM) | $ | 19,627 | $ | 20,597 | 104.9 | % | 110.80 | % | ||||||
Annual Total | 132.90 | %(1) | ||||||||||||
| 2009 | EPS | $ | 1.70 | $ | 1.89 | 111.2 | % | 146.00 | % | |||||
Sales ($=MM) | $ | 19,199 | $ | 19,537 | 101.8 | % | 101.80 | % | ||||||
Annual Total | 123.90 | % | ||||||||||||
Adjusted Annual Total(2) | 117.55 | %(2) | ||||||||||||
| (2) |
Special 2008-2010 Award:To drive continued cost reductions and productivity improvements through 2010, we granted a special, one-time long-term performance share unit award in 2008 to all executives worldwide. At the time of grant, approximately 100 of our top executives, including each of our Named Executive Officers, were eligible for long-term performance share unit awards on an annual basis. For this group,
the special award was granted in addition to athe regular 2008-2010 award and was granted in the same amount as the regular award. In addition, approximately 400 other executives who were not eligible for annual long-term performance share unit awards at the time of grant received this special 2008-2010 award. The special award has the same payout schedule as the regular 2008-2010 award described above. The payout of the special award is tied to the extent that annual cash flow and pretax profit margin goals are achieved over the 2008-2010 performance cycle. The two performance measuresmetrics are equally weighted. As described earlier, the company used cash flow as a performance measure for the first time in 2008. In that initial year,2008, cash flow was expressed in absolute dollar terms. In 2009 and 2010, the cash flow performance goal was measured as working capital plus capital expenditures expressed as a percent of net sales.
Below are Under the company’s2010 cash flow metric, achievement of 80% of target will yield a payout of 32.5% of target for that portion of the award; achievement below 80% of target will yield no payout; and achievement levels of 115% or higher of target will yield a maximum payout of 167.5% for that portion of the award. Under the 2010 pretax profit margin metric, achievement of 80% of target will yield a payout of 40% of target for that portion of the award; achievement below 80% of target will yield no payout; and achievement levels of 115% or higher of target will yield a maximum payout of 165% for that portion of the award. The 2008-2010 special award is payable at 146.7% of target award. The following table summarizes the performance and banked payout results for the first two years of the 2008-2010 specialrelating to this award:
Year | Measure | Target | Actual | % Achieve | % Payout | Measure | Target | Actual | % of Target | % Payout | ||||||||||||||||||||||||
| 2008 | Cash Flow ($=MM) | $ | 3,333 | $ | 3,852 | 115.6 | % | 165.00 | % | Cash Flow ($=MM) | $ | 3,333 | $ | 3,852 | 115.6 | % | 165.00 | % | ||||||||||||||||
Pretax Margin (%) | 18.8 | % | 20.7 | % | 110.1 | % | 139.20 | % | Pretax Margin (%) | 18.8 | % | 20.7 | % | 110.1 | % | 139.20 | % | |||||||||||||||||
Annual Total | 152.10 | % | Annual Total | 152.10 | % | |||||||||||||||||||||||||||||
| 2009 | Work Cap + CapEx (%) | 18.1 | % | 15.9 | % | 112.2 | % | 151.00 | % | Work Cap + CapEx (%) | 18.1 | % | 15.9 | % | 112.2 | % | 151.00 | % | ||||||||||||||||
Pretax Margin (%) | 21.9 | % | 24.5 | % | 111.9 | % | 149.50 | % | Pretax Margin (%) | 21.9 | % | 24.5 | % | 111.9 | % | 149.50 | % | |||||||||||||||||
Annual Total | 150.25 | % | Annual Total | 150.25 | % | |||||||||||||||||||||||||||||
| 2010 | Work Cap + CapEx (%) | 12.5 | % | 9.6 | %(1) | 123.2 | % | 167.50 | % | |||||||||||||||||||||||||
Adjusted Annual Total(1) | Not Applicable | (1) | Pretax Margin (%) | 23.5 | % | 24.4 | %(1) | 103.8 | % | 108.00 | % | |||||||||||||||||||||||
Annual Total | 137.75 | % | ||||||||||||||||||||||||||||||||
| Three-Year Total | 146.70 | % | ||||||||||||||||||||||||||||||||
| (1) |
When this special long-term performance award is taken into consideration, the 2009 target long-term incentive mix of our Named Executive Officers is: 46% long-term performance awards; 41% stock options; and 13% restricted stock units. This results in a long-term incentive mix in which 87% is performance-based.
2009-2011 Award: The 2009-2011 award has the same payout schedule as both of the 2008-2010 awards. The award is based 50% on non-GAAP diluted earnings per share, 25% on non-GAAP net sales, net of foreign exchange, and 25% on working capital and capital expenditures as a percent of net sales. For each of these metrics, the payout schedule is the same as that used under the 2008-2010 awards.
Below are the company’s performance and banked payout results for the first yeartwo years of the 2009-2011 award:
Year | Measure | Target | Actual | % Achieve | % Payout | Measure | Target | Actual | % of Target | % Payout | ||||||||||||||||||||||||
| 2009 | EPS | $ | 1.70 | $ | 1.89 | 111.2 | % | 146.00 | % | EPS | $ | 1.70 | $ | 1.89 | 111.2 | % | 146.00 | % | ||||||||||||||||
Sales ($=MM) | $ | 19,199 | $ | 19,537 | 101.8 | % | 101.80 | % | Sales ($=MM) | $ | 19,199 | $ | 19,537 | 101.8 | % | 101.80 | % | |||||||||||||||||
Work Cap + CapEx (%) | 18.1 | % | 15.9 | % | 112.2 | % | 151.00 | % | Work Cap + CapEx (%) | 18.1 | % | 15.9 | % | 112.2 | % | 151.00 | % | |||||||||||||||||
Annual Total | 136.20 | % | Annual Total | 136.20 | % | |||||||||||||||||||||||||||||
Adjusted Annual Total(1) | 133.18 | %(1) | Adjusted Annual Total | 133.18 | %(1) | |||||||||||||||||||||||||||||
| 2010 | EPS | $ | 2.17 | $ | 2.22 | (2) | 102.3 | % | 109.92 | % | ||||||||||||||||||||||||
| Sales Growth (%) | 6.1 | % | 4.1 | %(2) | 67.2 | % | 90.80 | % | ||||||||||||||||||||||||||
| Work Cap + CapEx (%) | 12.5 | % | 9.6 | %(2) | 123.2 | % | 167.50 | % | ||||||||||||||||||||||||||
| Annual Total | 119.54 | % | ||||||||||||||||||||||||||||||||
| (1) | Reflects the Committee’s use of negative discretion. Our long-term performance award program provides that discontinued operations be excluded from financial results for payout purposes. Therefore, the financial measures excluded the performance of the Mead Johnson business unit, which was split-off from |
| (2) | Actuals exclude the impact of the acquisition of ZymoGenetics and the portion of the impact of health care reform that could not be quantified at the time targets were approved by the Committee. The impact of health care reform was a decrease in net sales of $155 million and a decrease in non-GAAP diluted earnings per share of $0.055. The impact of ZymoGenetics was an increase in net sales of $15 million and a decrease in non-GAAP diluted earnings per share of $0.01. |
2010-2012 Award: The 2010-2012 award has the same performance metrics and weights, and the same performance goals and payout schedules as the 2009-2011 award: non-GAAP diluted earnings per share (weighted 50%), non-GAAP net sales growth, net of foreign exchange (weighted 25%) and working capital plus capital expenditures as a percent of net sales (weighted 25%). Below are performance and payout results for the first year of the 2010-2012 award:
Year | Measure | Target | Actual | % of Target | % Payout | |||||||||||||
2010 | EPS | $2.17 | $2.22(1) | 102.3% | 109.92% | |||||||||||||
| Sales Growth (%) | 6.1 | % | 4.1%(1) | 67.2% | 90.80% | |||||||||||||
| Work Cap + CapEx (%) | 12.5 | % | 9.6%(1) | 123.2% | 167.50% | |||||||||||||
| Annual Total | 119.54% | |||||||||||||||||
| (1) | Actuals exclude the impact of the acquisition of ZymoGenetics and the portion of the impact of health care reform that could not be quantified at the time targets were approved by the Committee. The impact of health care reform was a decrease in net sales of $155 million and a decrease in non-GAAP diluted earnings per share of $0.055. The impact of ZymoGenetics is an increase in net sales of $15 million and a decrease in non-GAAP diluted earnings per share of $0.01. |
Market Share Units
Market share units, granted for the first time in 2010, will vest 25% per year over a four-year period. On each vesting date, a payout factor is derived as a ratio of stock price on the vesting date (a 10-day average closing price) divided by stock price on the grant date (also a 10-day average). The payout factor is applied to the target number of units vesting on a given date, inclusive of accrued dividend equivalents, to determine the total number of units and dividend equivalents payable. No payout may exceed 200% of the target units payable (inclusive of accrued dividend equivalents). If the vesting-date stock price falls below 60% of the grant price, the target units, inclusive of the accrued dividend equivalents, are forfeited.
Stock Options and Restricted Stock Units: Stock
In 2010, we did not grant any stock options, were grantednor did we grant any service-based restricted stock units to executives as anpart of our annual long-term incentive program. Restricted stock units were only granted selectively to create long-term stockholder value. The sizeexecutives for purposes of the annual award an executive received was tied to a combination of grade levelattracting, retaining and individual performance. Within each grade level, we established a guideline award. On an individual level, the executive received more than or less than the award guideline, or potentially no award at all, based upon the executive’s performance as described earlier in the “Performance Management System” section on page 31.
The annual stock option awards granted to theproviding special recognition. Among our Named Executive Officers, and to other senior executives included a performance-based exercise threshold. To satisfy the exercise threshold, our common stock must close at a price at least 15% above the option exercise price for at least seven consecutive trading days. Although the options vest according to a normal vesting schedule (25% per year over four years), an executive may not exercise the options unless and until the exercise threshold is satisfied. Therefore, the executive will not realize any value related to these options until the stock price appreciates at least 15%. This threshold strengthened the link between the value realized by the executive and the creation of value for stockholders.
Restricted Stock Units: WeCommittee granted service-basedone special restricted stock unit awardsaward in 2010. Mr. Hooper received an award on May 3, 2010 valued at $500,000 to assist ushelp retain him and to recognize a significant increase in responsibility. Mr. Hooper assumed general management responsibility for Japan and several markets in the Asia/Pacific region while retaining key talent. Annual grants of restricted stock unitsresponsibility for Named Executive Officersthe U.S., Canada, Latin America and several other senior managementmarkets. Mr. Hooper’s award will vest one-third per year at the end ofon the third, fourth and fifth anniversaries following the date of grant. The size of the annual award an executive received was determined in the same manner as that described above for stock option awards.
In 2007, we shifted a portion of the dollar value previously allocated to restricted stock units to stock options and long-term performance awards so that time-based restricted stock units would only comprise approximately 15% of the target long-term program design for Named Executive Officers and other senior executives. In 2008 and 2009, we continued this practice to ensure that approximately 85% of our long-term program remained performance-based for our senior management team.
2009 Grants of Stock Options and Restricted Stock Units to our NEOs
To further enhance the link between executive pay and the company’s performance against its business strategy, the size of each Named Executive Officer’s stock option grant and restricted stock unit award was determined in large part based on the Committee’s assessment of the executive’s performance against objectives that drive our business strategy and demonstration of behaviors consistent with the BMS Commitment and the Core BMS Behaviors under the performance management system. In March of 2009, the Named Executive Officers received stock option grants and restricted stock unit awards ranging from 100% to 110% of the established guidelines for their respective grade levels. These award levels recognized management’s continued successful execution of the company’s biopharmaceutical business strategy and the individual performance described in the company’s 2009 Proxy Statement.date.
In addition, to recognize him for his continued achievement in creating a top-tier R&D pipeline, Dr. Sigal received a special one-time restricted stock unit award on March 3, 2009 valued at $2 million. When taking this special award into consideration, Dr. Sigal’s total compensation was positioned at slightly below the 75th percentile of his peer group in 2009.
Pursuant to Mr. Huet’s voluntary separation from the company, he forfeited all unvested performance share units, stock options and restricted stock units that were awarded to him since joining the company (see Termination of Employment Obligations Table for details).
OTHER ELEMENTS OF 20092010 COMPENSATION
In addition to the components set forth above, our senior executives, including all of our Named Executive Officers, were entitled to receive benefits in 2010 under the following benefits in 2009:plans or arrangements:
Post-Employment Benefits
Qualified and Non-Qualified Pension Plans
Qualified and Non-Qualified Savings Plans
Annual Incentive Deferral Plan
Severance Plan
Change-in-Control Arrangements
Other Compensation
General Employee Benefits
Post-Employment Benefits
We offer certain plans which provide compensation and benefits to employees who have terminated their employment. These plans are periodically reviewed by the Committee to ensure that they are consistent with competitive practice. The plans offered are common within our primary peer group and enhance our ability to attract and retain key talent.
Defined Benefit Pension Plans
Our defined benefit plans provide income for employees following retirement. The Retirement Income Plan is a tax-qualified plan, as defined under IRS regulations, and the Benefit Equalization Plan–Retirement Plan is a non-qualified plan that provides pension benefits above those allowed under the contribution limits for tax-qualified plans. All U.S. employees are eligible to participate in both defined benefit plans. The Summary Compensation Table reflects the annual increase in the actuarial value of these benefits. Current accrued benefits for each of the Named Executive Officers are provided in the Pension Benefit Table. (See “Program ChangesAs of December 31, 2009, we discontinued service accruals under our qualified and non-qualified pension plans in the U.S. and Puerto Rico for 2010” section on page 42 for a descriptionactive plan participants, including all of changesour Named Executive Officers, and we stopped adding new participants to our plans. For active plan participants at year-end 2009, we are allowing five additional years of pay growth in our pension plans.) These actions were taken to align our retirement program with our new biopharmaceutical business strategy and culture, to respond to the competitiveness of a changing industry, and to meet the mobility and career expectations of an evolving workforce.
Savings Plans
Our savings plans allow employees to defer a portion of their base salary and to receive matching contributions from the companyBMS to supplement their income in retirement. The Savings and Investment Program is a tax-qualified 401(k) plan, as defined under IRS regulations, and the Benefit Equalization Plan–Savings Plan is a non-qualified deferred compensation plan that allows employees to defer a portion of their base salary and to receive matching contributions from the companyBMS in excess of the contributions allowed under the Savings and Investment Program. The savings plans are designed to allow employees to accumulate savings for retirement on a tax-advantaged basis. All U.S. employees are eligible to participate in both savings plans. The Summary Compensation Table reflects company contributions to these plans during 2009,2010 in the “All Other Compensation” column. The Non-Qualified Deferred Compensation Table provides more detail on the BenefitsBenefit Equalization Savings Plan. (See “Program ChangesAs of January 1, 2010, we enhanced our qualified and non-qualified savings plans in the U.S. and Puerto Rico in order to maintain competitive retirement benefits in light of the pension plan phase-out described above. Listed below are the enhancements to the U.S. savings plans, in which the Named Executive Officers participate:
Recognition of total cash compensation for 2010” sectionthe purpose of employee and company contributions. Previously, the plans recognized base salary only.
Increase of the company match from 75% to 100% on page 42 forthe first 6% of total cash.
An additional automatic company contribution that is based on a descriptionpoint system of changes to our savings plans.)one’s age plus service: below 40 points, the automatic contribution is an additional 3% of total cash; between 40 and 60 points, the contribution is 4.5%; and at 60 points and above, the contribution is 6%. For those employees with 60 or more points who had 10 or more years of
service at year-end 2009, an additional automatic contribution of 2% is provided for a five-year period. Each Named Executive Officer has earned over 60 points and has more than 10 years of service. |
Annual Incentive Deferral Plan
We maintain a non-qualified deferred compensation program which is available to allplan for our executives, of the company including the Named Executive Officers. The program provides a benefit to eligible employees by allowing them to defer taxes on earned compensation. This is a common program withinUntil we discontinued new deferrals under the company’s competitive peer group. Underplan, effective January 1, 2010, the program,plan permitted executives are permitted to defer up to 100% of their annual cash incentive awards into a choice of two funds: a Bristol-Myers Squibb common stock unit fund and a U.S. Treasury Bill fund. Although we no longer permit new deferrals under the plan, we maintain the plan for executives who made deferrals prior to 2010. We do not pay above-market interest rates on these
investments. Upon retirement or termination, the employee isplan participants are eligible to receive thetheir deferred amounts based on a previously-selected payout schedule. The Committee may approve accelerated distributions in the event of an unforeseeable emergency. In 2009, none of the Named Executive Officers deferred their annual incentive award payments. The Non-Qualified Deferred Compensation Table provides more detail on this programplan for those Named Executive Officers who participated in previous years. The rationale for discontinuing new deferrals under the plan is that (i) very few employees elected to defer their bonuses in previous years. (See “Program Changes for 2010” section on page 42 for a description of changes toprior years; and (ii) partial bonus deferrals are now possible under our annual incentive deferral plan.)enhanced qualified and non-qualified savings plans.
Severance Plan
The Bristol-Myers Squibb Senior Executive Severance Plan provides a competitive level of severance protection for certain senior executives to help us attract and retain key talent necessary to run our company. This plan covers 14 executives, including all of the Named Executive Officers except Mr. Andreotti, who is covered under a separate arrangement. Mr. Andreotti was formerly an Italian employee who was covered by the national collective agreement for Italian executives. Under this agreement, the company and Mr. Andreotti negotiated certain benefits, including severance benefits. When, at the request of the company, Mr. Andreotti became a U.S. employee, we agreed to provide Mr. Andreotti with severance benefits comparable to the benefits to which he was entitled as an Italian employee.1Officers. The value of this benefit for our Named Executive Officers is shown in the “Post-Termination Benefits” section on page 55.57.
Change-in-Control Arrangements
We have entered into change-in-control agreements with certain executives including all of the CEO and other Named Executive Officers. These agreements enable management to evaluate and support potential transactions that might be beneficial to stockholders even though the result would be a change in control of the company.BMS. Additionally, the agreements provide for continuity of management in the event of a change in control. Our agreements require a “double-trigger” before any payments are made to an executive. This means that payments are only made in the event of a change in control and subsequent involuntary termination or termination for good reason of the employee within three years after a change in control.
Consistent with competitive practice, With respect to our Named Executive Officers, if payments made to a covered officer are subject to excise tax as excess parachute payments by the Internal Revenue Code, we will gross up the compensation to fully offset the excise taxes. However, if the payment does not exceed the excise tax threshold by more than 10%, we will reduce the payment so that no portion of the payment is subject to excise tax and no gross-up would be made. We believe that this is a best practice relating to gross-up provisions in change-in-control arrangements. Effective January 1, 2009, change-in-control agreements were amended for documentary compliance with Section 409A of the Internal Revenue Code. Agreements are automatically extended in one-year increments unless either the company or the executive gives prior notice of termination of the agreement or a change in control shall have occurred prior to January 1 of such year. If a change in control occurs during the term of the agreement, the agreement will continue in effect for a period of not less than 36 months beyond the month in which such change in control occurred. The value of this benefit for our Named Executive Officers is provided in the “Post-Termination Benefits” section.
The Committee reviews the benefits provided under the agreements on an annual basis to ensure they meet our needs and, with the assistance of its independent compensation consultant, ensure that the benefits are within competitive parameters. Given emerging competitive trends, the Committee made certain changes to the agreements effective September 1, 2010 for newly eligible executives as follows:
The valueprotection period for receiving change-in-control benefits has been reduced from three to two years following the effective date of this benefit for Named Executive Officers is provideda change in control.
We will not gross up compensation on excess parachute payments.
We will not provide any pension subsidy or enhancement. In lieu of such subsidy or enhancement, we will provide under the “Post-Termination Benefits” section on page 55.non-qualified savings plan a continuation of company matching contributions and automatic year-end contributions equal to the length of the severance period.
Other Compensation
Mr. Cornelius was provided with a company car and driver in 2009.during 2010 when he served as our CEO. Personal use of such company car and driver is disclosed in the All Other Compensation Detail table. WeMr. Cornelius was the only Named Executive Officer who received a perquisite in 2010. Since Mr. Cornelius’ retirement, we have eliminated all other executiveprovided no perquisites forto our Named Executive Officers and other senior executives.Officers.
|
TAX IMPLICATIONS OF EXECUTIVE COMPENSATION PROGRAM
Section 162(m) of the Internal Revenue Code includes potential limitations on the deductibility of compensation in excess of $1 million paid to certain Named Executive Officers. A significant portion of the compensation we pay to our Named Executive Officers qualifies as “performance-based compensation” for purposes of Section 162(m) and is, therefore, fully deductible by the companyBMS for federal income tax purposes. We view preserving tax deductibility as an important objective, but not the sole objective, in establishing executive compensation. In specific instances, we may authorize compensation arrangements that are not fully tax deductible, but which promote other important objectives of the company.objectives. The portions of compensation paid in 20092010 to our Named Executive Officers that are not tax deductible include: (a) distributions to Dr. Sigal and Messrs. Cornelius, Andreotti and Hooper pursuant to the vesting of restricted stock units as they are not deemed to be performance-based awards (see Grants of Plan-Based Awards Table and Option Exercises and Stock Vesting Table); and (b) salary amounts in excess of $1 million paid to Dr. Sigal and Messrs. Cornelius andMr. Andreotti (see Summary Compensation Table).
There may be a lossUnder the 2008 portion of deductibility on the payout of our 2008-2010 special long-term performance share unit award, in 2011 due to the establishment of acash flow metric under the award that did not qualify for deductibility. As a result, we lost deductibility under Section 162(m).
As mentioned earlier,on payments totaling $2.1 million. Under the 2009 portions of the 2008-2010 performance share unit award and the 2008-2010 special performance share unit award, certain 2009 expenses related to the acquisition of Medarex, Inc. and the extension of our agreement with Otsuka to market Abilify® in the U.S. were excluded from the calculation of payouts under our compensation programs.after targets had been set by the Committee. As a result of this exclusion, there may be a loss of deductibility on the payout of the 2008-2010 regular long-term performance award in 2011 and the payout of the 2009-2011 long-term performance award in 2012. This exclusion also applied to the payout of the 2007-2009 long-term performance award in 2010, for which the companythese exclusions, we lost deductibility on payments totaling $2.4$3.8 million.
2010 MANAGEMENT CHANGES
As previously announced, on May 4, 2010, the date of the Annual Meeting, Mr. Cornelius will retire as Chief Executive Officer of the company. On March 2, 2010, the Board of Directors appointed Lamberto Andreotti as Chief Executive Officer–Designate to provide for a smooth transition. Mr. Andreotti will become the Chief Executive Officer of the company on May 4, 2010 and Mr. Cornelius will become the Non-Executive Chairman of the Board on that date. The Board of Directors has approved Mr. Andreotti’s new compensation package as CEO, effective May 4, 2010, as detailed below:
Mr. Andreotti’s salary will increase from $1,251,051 to $1,400,000, which represents an increase of 12%.
His annual target bonus will increase from 135% to 150% of base salary. Thus, his annual target total cash compensation will increase from $2,939,970 to $3,500,000, representing an increase of 19%.
The 2010 target value of Mr. Andreotti’s long-term incentives will increase from $2,874,999 to $7,500,000. Thus, his target total compensation (defined as target total cash compensation plus target long-term incentives value) will increase from $5,814,969 to $11,000,000, reflecting an increase of 89%.
Effective March 2, 2010, Mr. Andreotti received long-term incentive awards valued at $4,024,999, representing a grant at 140% of target. Consistent with the design of the new long-term incentive program for all executives, Mr. Andreotti received 60% of his March grant as performance share units (valued at $2,414,999) and 40% as market share units (valued at $1,610,000).
Effective May 4, 2010, Mr. Andreotti will receive additional long-term incentive awards valued at $3,475,001, which will bring his total value for 2010 to $7,500,000, the long-term incentive target for his new position. Again, he will receive 60% of the new grant as performance share units ($2,085,001) and 40% as market share units ($1,390,000).
In the event Mr. Andreotti is terminated involuntarily, not for cause and absent a change in control, he will be eligible to receive severance pay equal to two times his base salary. This is the same level of benefit available to all other Named Executive Officers. Mr. Andreotti will no longer be eligible to receive a special severance benefit that had entitled him to 2.99 times base salary plus target bonus.
There will be no change to Mr. Andreotti’s current severance benefits in the event of a change in control. His benefits are the same as those available to other Named Executive Officers.
Mr. Andreotti will not receive any company perquisites.
Mr. Andreotti’s new total compensation package is positioned at approximately the 25th percentile among CEOs within our current proxy peer group. The Committee believes Mr. Andreotti’s new compensation package positions him appropriately among his peers when taking multiple factors into consideration, including Mr. Andreotti’s new tenure as Chief Executive Officer, peers that hold the dual role of Chairman and CEO, and relative company size.
PROGRAM CHANGES FOR 20102011
The companyWe implemented certain changes to our compensation and benefitperformance management programs for 20102011 to continue driving the successful executionevolution of our biopharmaceutical business strategy.strategy and culture. These changes are described below:
| • |
|
| • |
|
| • | Performance Management Program: We are implementing a modified set of behaviors under our performance management program for 2011. The |
2011 BMS BioPharma Behaviors Decide and Act Connect and Collaborate |
| • |
|
|
|
Through the redesign of our long-term incentive program, we were able to achieve several objectives that support our biopharmaceutical business strategy:
The entire long-term incentive program for executives is performance-based.
The new design applies uniformly to all levels of executives, thus promoting organizational alignment with our biopharmaceutical business goals.
With payout opportunities spread over several years, including the payouts relating to dividend equivalents, the new program enhances our capacity to retain our executives.
The elimination of stock option awards and the reduction in long-term incentive targets will result in the new program being approximately 17% less costly than the current program.
The new program offers the opportunity to earn dividend equivalents upon payout of performance share units and provides a greater likelihood of delivering value to the executive.
We believe our new long-term incentive program serves the best interests of our stockholders by focusing the efforts of our executives on key financial drivers of long-term success and on total stockholder return while enabling the company to reduce expenses.
|
|
Increase of the company match from 75% to 100% on the first 6% of total cash. Total cash is defined as base salary plus annual bonus payments.
An additional automatic company contribution that is based on a point system of one’s age plus service: below 40 points, the automatic contribution is an additional 3% of total cash; between 40 and 60 points, the contribution is 4.5%; and at 60 points and above, the contribution is 6%. For those employees with 60 or more points who had 10 or more years of service at year-end 2009, an additional automatic contribution of 2% is provided for a five-year period.
|
CORPORATE POLICIES COVERING EXECUTIVE COMPENSATION
Share Ownership and Retention GuidelinesPolicy
In order to preserve the link between the interests of the Named Executive Officers and those of stockholders, executives are expected to use the shares acquired upon the exercise of their stock options, after satisfying the cost of exercise and taxes, to establish and maintain a significant level of direct ownership. This same expectation applies to shares acquired upon the vesting of (i) restricted stock units, and, beginning in 2010,(ii) market share units.units and (iii) performance share unit awards granted in 2011 and beyond. We continue to maintain longstanding share
ownershipshare-ownership expectations for our senior executives. Our guidelines require that the CEO hold shares with a value of eight times his base salary before he can sell any net shares following the exercise of options or the vesting of restricted stock units and market share units. The other Named Executive Officers must hold shares with a value of five times their base salary before they can sell any net shares following the exercise of options or the vesting of restricted stock units and market share units. Once these share ownership guidelines are attained, the executive must retain 75% of all shares acquired, net of shares withheld for tax purposes, following any subsequent option exercises or vesting of restricted stock units and market share units for a period of one year. All executives were in compliance withCommittee revised our share ownership and retention guidelines during 2009, except that the Committee permitted Mr. Corneliuspolicy in 2010 to tender sharessubject all forms of Bristol-Myers Squibb stock for shares of Mead Johnson Nutrition Company stock pursuantequity grants to the company exchange offer. The Committee determined that despite Mr. Cornelius not having attainedownership and retention requirements and to bring the sharepolicy in closer alignment with competitive practice. Our Named Executive Officers must comply with the following ownership guidelines, it was appropriate for Mr. Cornelius to participate in the exchange offer and increase his ownership of Mead Johnson shares as Mead Johnson’s Chairman of the Board. Mr. Cornelius has since met the company’s share ownership requirements.retention requirements:
| Stock Ownership Guideline as a Multiple of Salary | Share Retention Policy - applied to all shares acquired, net of taxes | 2010 Compliance with Share Ownership and Retention Policy | ||||||||||||||
Executive | ||||||||||||||||
| Prior to Achieving Guideline | After Achieving Guideline | |||||||||||||||
L. Andreotti | 6 x | 100 | % | 75% for 1 year | Yes | |||||||||||
C. Bancroft | 3 x | 100 | % | 75% for 1 year | Yes | |||||||||||
E. Sigal | 3 x | 100 | % | 75% for 1 year | Yes | |||||||||||
A. Hooper | 3 x | 100 | % | 75% for 1 year | Yes | |||||||||||
S. Leung | 2 x | 100 | % | 75% for 1 year | Yes | |||||||||||
Recoupment of Compensation
We maintain clawback provisions relating to stock options, restricted stock units, long-term performance awards and, beginning in 2010, market share units. Under these clawback provisions, executives that violate non-competition or non-solicitation agreements, or otherwise act in a manner detrimental to the company’sour interests, forfeit any outstanding awards, and any accrued and unpaid dividend equivalents underlying these awards, as of the date such violation is discovered and have to return any gains realized in the twelve months prior to the violation. These provisions serve to protect our intellectual property and human capital, and help ensure that executives act in the best interest of the companyBMS and itsour stockholders.
In 2005, the Board adopted a policy wherein the Board will seek reimbursement of annual incentives paid to an executive if such executive engaged in misconduct that caused or partially caused a restatement of financial results. In such an event, the companywe will seek to claw back the executive’s entire annual incentive for the relevant period, plus a reasonable rate of interest. This policy may be viewed on our website at www.bms.com.
We are awaiting final rules from the Securities and Exchange Commission to implement Dodd-Frank legislation on clawback provisions, and will revise our policy, as appropriate, based on such rules.
Equity Grant Policy
In 2006, the Committee approved a policy covering equity grants to all employees. For the Named Executive Officers, the policy is as follows.
Approval of Awards
Awards granted to the CEO must be approved by the Committee and recommended by the Committee to and approved by at least 75% of the independent directors of the Board.
The Committee must approve awards to all Named Executive Officers.
Grant Effective Date
Annual Awards
For regularly-scheduled annual awards, the grant effective date is the date in March on which the Compensation and Management Development Committee and full Board meet.
All Other Awards
For awards granted to current employees at any other time during the year, the grant effective date is the first business day of the month following the approval date, except that if the approval date falls on the first business day of a given month, the grant effective date is the approval date.
For awards granted to new hires, the grant effective date is the first business day of the month following the employee’s hire date, except that if the employee’s hire date falls on the first business day of a given month, the grant effective date is the employee’s hire date.
In no case whatsoever will the grant effective date precede the approval date of a given award.
Grant Price
The grant price of awards granted prior to January 1,March 2, 2010 was the closing price on the date of grant (i.e., the Fair Market Value as defined in our 2007 Stock Award and Incentive Plan).
The grant price of awards granted on or after January 1,March 2, 2010, with the exception of stock options, will beis a 10-day average closing price (i.e., an average of the closing price on the grant date plus the nine prior trading days). For stock options that may be granted under special circumstances, the grant price will be the closing price on the date of grant.
Policy Against the Repricing of Stock Options
We have always maintained a consistent policy against the repricing of stock options. We believe this is a critical element in maintaining the integrity of the equity compensation program and ensuring alignment of senior executives’ interests with the interests of stockholders. The Board of Directors has adopted a formal policy prohibiting the repricing of stock options. This policy may be viewed on the company’sour website at www.bms.com.
Policy Regarding Stockholder Approval of Severance
The Board has approved a policy that requires stockholder approval of any future agreements that provide for cash severance payments in excess of 2.99 times the sum of an executive’s base salary plus annual incentive. “Cash severance payments” exclude accrued incentive payments, the value of equity acceleration, benefits continuation or the increase in retirement benefits triggered by severance provisions or tax gross-up payments. This policy may be viewed on the company’sour website at www.bms.com.
Compensation and Management Development Committee Report
The Compensation and Management Development Committee (Committee) of Bristol-Myers Squibb Company has reviewed and discussed with management the Compensation Discussion and Analysis on pages 2526 to 4546 of this Proxy Statement as required under Item 402(b) of Regulation S-K. Based on its review and discussions with management, the Committee recommended to the company’s full Board that the Compensation Discussion and Analysis be included in this Proxy Statement.
Compensation and Management Development Committee
Lewis B. Campbell, Chair
Michael Grobstein
Leif Johansson
Vicki L. Sato, Ph.D.
Togo D. West, Jr.
The following tables and notes present the compensation provided to Lamberto Andreotti, Chief Executive Officer, Charles A. Bancroft, Chief Financial Officer, James M. Cornelius, our former Chairman and& Chief Executive Officer, Jean-Marc Huet, our former Executive Vice President and Chief Financial Officer as well as the three other most highly compensated Executive Officers.
Summary Compensation Table
forFor Fiscal Year Ended December 31, 2010, 2009 2008, and 20072008
Name and | Year | Salary (1) | Bonus (2) | Stock Awards (3) | Option Awards (4) | Non-Equity Incentive Plan Compen- sation (5) | Change in Pension Value and Non- Qualified Deferred Compen- sation Earnings (6) | All Other Compen- sation (7) | Total | |||||||||||||||||
James M. Cornelius | 2009 | $ | 1,500,000 | — | $ | 5,661,943 | $ | 5,209,633 | $ | 4,494,825 | $ | 1,218,670 | $ | 136,363 | $ | 18,221,434 | ||||||||||
Chairman & Chief Executive Officer | 2008 | $ | 1,488,077 | — | $ | 5,122,554 | $ | 7,146,853 | $ | 4,475,000 | $ | 1,341,903 | $ | 136,309 | $ | 19,710,697 | ||||||||||
| 2007 | $ | 1,352,115 | $ | 1,062,500 | $ | 3,148,783 | $ | 3,352,266 | $ | 2,220,801 | $ | 914,247 | $ | 424,954 | $ | 12,475,667 | ||||||||||
Jean-Marc Huet(8) | 2009 | $ | 913,361 | — | $ | 1,193,867 | $ | 1,350,155 | $ | 0 | $ | 250,633 | $ | 42,908 | $ | 3,750,924 | ||||||||||
Former EVP & CFO | 2008 | $ | 675,000 | $ | 100,000 | $ | 924,598 | $ | 1,392,111 | $ | 1,583,993 | $ | 171,800 | $ | 285,736 | $ | 5,133,238 | |||||||||
Lamberto Andreotti(9) | 2009 | $ | 1,244,730 | — | $ | 1,636,206 | $ | 1,684,655 | $ | 3,076,402 | $ | 1,307,315 | $ | 56,013 | $ | 9,005,321 | ||||||||||
President & Chief Operating Officer | 2008 | $ | 1,211,141 | — | $ | 1,340,374 | $ | 1,812,847 | $ | 2,676,668 | $ | 887,568 | $ | 57,801 | $ | 7,986,400 | ||||||||||
| 2007 | $ | 1,098,637 | — | $ | 881,942 | $ | 1,657,056 | $ | 1,866,344 | $ | 550,550 | $ | 111,922 | $ | 6,166,451 | |||||||||||
Elliott Sigal, M.D., Ph.D. | 2009 | $ | 1,014,846 | — | $ | 3,436,891 | $ | 1,350,155 | $ | 2,189,879 | $ | 1,966,999 | $ | 45,668 | $ | 10,004,439 | ||||||||||
EVP BMS CO CSO | 2008 | $ | 985,845 | — | $ | 1,340,374 | $ | 1,812,847 | $ | 2,084,324 | $ | 1,579,640 | $ | 49,449 | $ | 7,852,480 | ||||||||||
& President R&D | 2007 | $ | 880,281 | — | $ | 904,225 | $ | 1,726,100 | $ | 1,430,387 | $ | 1,077,995 | $ | 112,772 | $ | 6,131,760 | ||||||||||
Anthony C. Hooper | 2009 | $ | 782,308 | — | $ | 1,005,715 | $ | 1,022,271 | $ | 1,113,373 | $ | 1,068,498 | $ | 35,204 | $ | 5,027,369 | ||||||||||
President, Americas | 2008 | $ | 693,464 | — | $ | 805,287 | $ | 1,016,297 | $ | 1,065,861 | $ | 798,823 | $ | 32,591 | $ | 4,412,323 | ||||||||||
| 2007 | $ | 643,890 | — | $ | 885,758 | $ | 738,559 | $ | 681,660 | $ | 502,710 | $ | 49,371 | $ | 3,501,948 | |||||||||||
Name and Principal Position | Year (1) | Salary (2) | Bonus | Stock Awards (3) | Option Awards (4) | Non-Equity Incentive Plan Compen- sation (5) | Change in Pension Value and Non- Qualified Deferred Compen- sation Earnings (6) | All Other Compen- sation (7) | Total | |||||||||||||||||||||||||||
Lamberto Andreotti | 2010 | $ | 1,347,868 | — | $ | 5,808,704 | $ | 0 | $ | 3,131,544 | $ | 863,366 | $ | 619,398 | $ | 11,770,880 | ||||||||||||||||||||
Chief Executive Officer | 2009 | $ | 1,244,730 | — | $ | 1,636,206 | $ | 1,684,655 | $ | 3,076,402 | $ | 1,307,315 | $ | 56,013 | $ | 9,005,321 | ||||||||||||||||||||
| 2008 | $ | 1,211,141 | — | $ | 1,340,374 | $ | 1,812,847 | $ | 2,676,668 | $ | 887,568 | $ | 57,801 | $ | 7,986,400 | |||||||||||||||||||||
Charles A. Bancroft | 2010 | $ | 678,830 | — | $ | 1,000,385 | $ | 0 | $ | 978,595 | $ | 1,418,419 | $ | 142,409 | $ | 4,218,638 | ||||||||||||||||||||
Chief Financial Officer | ||||||||||||||||||||||||||||||||||||
Elliott Sigal, M.D., Ph.D. | 2010 | $ | 1,020,000 | — | $ | 2,918,790 | $ | 0 | $ | 1,952,642 | $ | 1,941,559 | $ | 449,383 | $ | 8,282,374 | ||||||||||||||||||||
EVP BMS CO CSO & President R&D | 2009 | $ | 1,014,846 | — | $ | 3,436,891 | $ | 1,350,155 | $ | 2,189,879 | $ | 1,966,999 | $ | 45,668 | $ | 10,004,439 | ||||||||||||||||||||
| 2008 | $ | 985,845 | — | $ | 1,340,374 | $ | 1,812,847 | $ | 2,084,324 | $ | 1,579,640 | $ | 49,449 | $ | 7,852,480 | |||||||||||||||||||||
Anthony C. Hooper | 2010 | $ | 800,000 | — | $ | 2,649,962 | $ | 0 | $ | 1,276,236 | $ | 1,154,442 | $ | 267,872 | $ | 6,148,512 | ||||||||||||||||||||
SVP Commercial Operations | 2009 | $ | 782,308 | — | $ | 1,005,715 | $ | 1,022,217 | $ | 1,113,373 | $ | 1,068,498 | $ | 35,204 | $ | 5,027,315 | ||||||||||||||||||||
& President US, Japan and Intercontinental | 2008 | $ | 693,464 | — | $ | 805,287 | $ | 1,016,297 | $ | 1,065,861 | $ | 798,823 | $ | 32,591 | $ | 4,412,323 | ||||||||||||||||||||
Sandra Leung | 2010 | $ | 675,000 | — | $ | 1,824,035 | $ | 0 | $ | 797,648 | $ | 1,110,255 | $ | 222,965 | $ | 4,629,903 | ||||||||||||||||||||
General Counsel | ||||||||||||||||||||||||||||||||||||
& Corporate Secretary | ||||||||||||||||||||||||||||||||||||
James M. Cornelius | 2010 | $ | 525,000 | — | $ | 4,543,486 | $ | 0 | $ | 1,495,709 | -$ | 3,639,528 | $ | 628,838 | $ | 3,553,506 | ||||||||||||||||||||
Former Chairman & CEO | 2009 | $ | 1,500,000 | — | $ | 5,661,943 | $ | 5,209,633 | $ | 4,494,825 | $ | 1,218,670 | $ | 136,363 | $ | 18,221,434 | ||||||||||||||||||||
| 2008 | $ | 1,488,077 | — | $ | 5,122,554 | $ | 7,146,853 | $ | 4,475,000 | $ | 1,341,903 | $ | 136,309 | $ | 19,710,697 | |||||||||||||||||||||
| (1) | For Mr. Bancroft and Ms. Leung, compensation is not shown for fiscal years 2008 and 2009 because they were not named executive officers in those fiscal years. |
| (2) | Reflects actual salary earned. For |
| (3) | Represents aggregate grant date fair value under |
| Performance Share Units | Market Share Units | ||||||||||||||||||||||||
Name | 2007 | 2008 | 2009 | 2008 | 2009 | 2010 | 2010 | ||||||||||||||||||
James M. Cornelius | $ | 3,141,563 | $ | 6,007,660 | $ | 7,086,645 | |||||||||||||||||||
Jean-Marc Huet | n.a. | $ | 921,736 | $ | 1,251,147 | ||||||||||||||||||||
Lamberto Andreotti | $ | 763,717 | $ | 1,554,904 | $ | 1,936,847 | $ | 1,554,904 | $ | 1,936,847 | $ | 4,659,199 | $ | 6,049,061 | |||||||||||
Elliott Sigal, M.D., Ph.D. | $ | 763,717 | $ | 1,554,904 | $ | 1,785,818 | $ | 1,554,904 | $ | 1,785,818 | $ | 2,890,966 | $ | 2,380,556 | |||||||||||
Anthony C. Hooper | $ | 394,721 | $ | 933,720 | $ | 1,185,985 | $ | 933,720 | $ | 1,185,985 | $ | 2,114,703 | $ | 1,760,649 | |||||||||||
James M. Cornelius | $ | 6,007,660 | $ | 7,086,645 | $ | 7,593,756 | n.a. | ||||||||||||||||||
Charles A. Bancroft (1) | n.a. | n.a. | $ | 739,907 | $ | 1,116,564 | |||||||||||||||||||
Sandra Leung (1) | n.a. | n.a. | $ | 1,836,770 | $ | 1,451,533 | |||||||||||||||||||
| (4) | Represents aggregate grant date fair value under |
| (5) | Represents |
| (6) | Includes increase in estimated value of accrued benefits during the year. The company does not pay above-market interest rates on deferred compensation. |
| (7) | Includes value of perquisites and company contributions to the Qualified and BEP |
forFor 2010, 2009, 2008, and 20072008
Name | Year | Car (1) | Air Travel (2) | Financial Counseling (3) | Tax Preparation (3) | Total Perquisites and Other | Company Contributions to Savings Plans | Total All Other Compensation | Year | Car (1) | Air Travel (2) | Financial Counseling (3) | Tax Preparation (3) | Total Perquisites and Other | Company Contributions to Savings Plans | Total All Other Compensation | ||||||||||||||||||||||||||||||||||||||||
James M. Cornelius | 2009 | $ | 68,863 | $ | 0 | $ | 0 | $ | 0 | $ | 68,863 | $ | 67,500 | $ | 136,363 | |||||||||||||||||||||||||||||||||||||||||
| 2008 | $ | 69,346 | $ | 0 | $ | 0 | $ | 0 | $ | 69,346 | $ | 66,963 | $ | 136,309 | ||||||||||||||||||||||||||||||||||||||||||
| 2007 | $ | 67,770 | $ | 84,636 | $ | 0 | $ | 0 | $ | 377,090 | (4) | $ | 47,864 | $ | 424,954 | |||||||||||||||||||||||||||||||||||||||||
Jean-Marc Huet | 2009 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 1,807 | (8) | $ | 41,101 | $ | 42,908 | ||||||||||||||||||||||||||||||||||||||||
| 2008 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 276,390 | (7) | $ | 9,346 | $ | 285,736 | |||||||||||||||||||||||||||||||||||||||||
Lamberto Andreotti | 2009 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 56,013 | $ | 56,013 | 2010 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 619,398 | $ | 619,398 | ||||||||||||||||||||||||||
| 2008 | $ | 0 | $ | 0 | $ | 0 | $ | 3,300 | $ | 3,300 | $ | 54,501 | $ | 57,801 | 2009 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 56,013 | $ | 56,013 | |||||||||||||||||||||||||||
| 2007 | $ | 14,105 | $ | 35,953 | $ | 8,696 | $ | 3,000 | $ | 62,483 | (6) | $ | 49,439 | $ | 111,922 | 2008 | $ | 0 | $ | 0 | $ | 0 | $ | 3,300 | $ | 3,300 | $ | 54,501 | $ | 57,801 | ||||||||||||||||||||||||||
Charles A. Bancroft | 2010 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 142,409 | $ | 142,409 | |||||||||||||||||||||||||||||||||||||||||
Elliott Sigal, M.D., Ph.D. | 2009 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 45,668 | $ | 45,668 | 2010 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 449,383 | $ | 449,383 | ||||||||||||||||||||||||||
| 2008 | $ | 0 | $ | 1,586 | $ | 0 | $ | 3,500 | $ | 5,086 | $ | 44,363 | $ | 49,449 | 2009 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 45,668 | $ | 45,668 | |||||||||||||||||||||||||||
| 2007 | $ | 13,250 | $ | 48,574 | $ | 8,340 | $ | 2,995 | $ | 73,159 | (5) | $ | 39,613 | $ | 112,772 | 2008 | $ | 0 | $ | 1,586 | $ | 0 | $ | 3,500 | $ | 5,086 | $ | 44,363 | $ | 49,449 | ||||||||||||||||||||||||||
Anthony C. Hooper | 2009 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 35,204 | $ | 35,204 | 2010 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 267,872 | $ | 267,872 | ||||||||||||||||||||||||||
| 2008 | $ | 0 | $ | 0 | $ | 0 | $ | 1,385 | $ | 1,385 | $ | 31,206 | $ | 32,591 | 2009 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 35,204 | $ | 35,204 | |||||||||||||||||||||||||||
| 2007 | $ | 8,514 | $ | 0 | $ | 8,075 | $ | 3,590 | $ | 20,396 | (6) | $ | 28,975 | $ | 49,371 | 2008 | $ | 0 | $ | 0 | $ | 0 | $ | 1,385 | $ | 1,385 | $ | 31,206 | $ | 32,591 | ||||||||||||||||||||||||||
Sandra Leung | 2010 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 222,965 | $ | 222,965 | |||||||||||||||||||||||||||||||||||||||||
James M. Cornelius | 2010 | $ | 26,459 | $ | 0 | $ | 0 | $ | 0 | $ | 26,459 | $ | 602,379 | $ | 628,838 | |||||||||||||||||||||||||||||||||||||||||
| 2009 | $ | 68,863 | $ | 0 | $ | 0 | $ | 0 | $ | 68,863 | $ | 67,500 | $ | 136,363 | ||||||||||||||||||||||||||||||||||||||||||
| 2008 | $ | 69,346 | $ | 0 | $ | 0 | $ | 0 | $ | 69,346 | $ | 66,963 | $ | 136,309 | ||||||||||||||||||||||||||||||||||||||||||
| (1) | Reflects the value of a company car and |
| (2) |
| (3) | Reflects bills paid for financial counseling and tax preparation services. For 2008, reflects the bills paid for 2007 tax preparation services. These benefits were discontinued on 12/31/2007. |
20092010 Fiscal Year
Name | Grant Date (1) | Estimated Future Payouts Under Non-Equity Incentive Plan Awards(2) | Estimated Future Payouts Under Equity Incentive Plan Awards (shares) | All Other Stock Awards: # of Shares of Stock or Units (7) | All Other Option Awards: # of Securities Underlying Options (8) | Exercise or Base Price of Option Awards ($/ Share) (9) | Grant Date Fair Value of Stock and Option Awards (10) | Grant Date(1) | Approval Date (1) | Estimated Future Payouts Under Non-Equity Incentive Plan Awards(2) | Estimated Future Payouts Under Equity Incentive Plan Awards (shares) | All Other Stock Awards: # of Shares of Stock or Units | All Other Option Awards: # of Securities Under- lying Options | Exercise or Base Price of Option Awards ($/Share) | Grant Date Fair Value of Stock and Option Awards (3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Threshold ($) | Target ($) | Maximum ($) | Threshold (#) | Target (#) | Maximum (#) | Threshold ($) | Target ($) | Maximum ($) | Threshold (#) | Target (#) | Maximum (#) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
James M. Cornelius | $ | 731,250 | $ | 2,250,000 | $ | 5,568,750 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Name | Grant Date(1) | Approval Date (1) | Threshold ($) | Target ($) | Maximum ($) | Threshold (#) | Target (#) | Maximum (#) | All Other Stock Awards: # of Shares of Stock or Units | All Other Option Awards: # of Securities Under- lying Options | Exercise or Base Price of Option Awards ($/Share) | Grant Date Fair Value of Stock and Option Awards (3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 12,750 | 56,667 | 124,667 | (3) | $ | 938,972 | 2,383 | 14,667 | 24,567 | (4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 11,333 | 56,667 | 93,501 | (4) | $ | 888,539 | 2,383 | 14,667 | 24,384 | (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 11,333 | 56,667 | 93,501 | (5) | $ | 888,539 | 03/02/10 | 2,136 | 26,271 | 44,004 | (6) | $ | 582,165 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 8,530 | 85,303 | 140,750 | (6) | $ | 1,265,897 | 03/02/10 | 2,658 | 32,697 | 54,767 | (7) | $ | 808,270 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 95,945 | $ | 1,679,997 | 05/04/10 | 03/02/10 | 2,275 | 27,979 | 46,865 | (7a) | $ | 707,029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 1,140,188 | $ | 17.51 | $ | 5,209,633 | 03/02/10 | 39,236 | 65,394 | 130,788 | (8) | $ | 1,613,270 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Jean-Marc Huet(11) | $ | 356,265 | $ | 1,096,200 | $ | 2,713,095 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,933 | 14,667 | 24,201 | (4) | $ | 229,979 | 05/04/10 | 03/02/10 | 33,575 | 55,958 | 111,916 | (8a) | $ | 1,411,261 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,933 | 14,667 | 24,201 | (5) | $ | 229,979 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,010 | 20,102 | 33,168 | (6) | $ | 298,314 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 24,877 | $ | 435,596 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 295,497 | $ | 17.51 | $ | 1,350,155 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lamberto Andreotti | $ | 536,240 | $ | 1,649,970 | $ | 4,083,676 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 3,300 | 14,667 | 32,267 | (3) | $ | 243,032 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Charles A. Bancroft | $ | 61,613 | $ | 662,500 | $ | 1,617,726 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,933 | 14,667 | 24,201 | (4) | $ | 229,979 | 03/02/10 | 342 | 2,104 | 3,524 | (4) | $ | 49,255 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,933 | 14,667 | 24,201 | (5) | $ | 229,979 | 03/02/10 | 342 | 2,104 | 3,498 | (5) | $ | 49,255 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,627 | 26,270 | 43,346 | (6) | $ | 389,847 | 03/02/10 | 234 | 2,883 | 4,829 | (6) | $ | 63,887 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 31,032 | $ | 543,370 | 03/02/10 | 920 | 11,315 | 18,953 | (7) | $ | 279,707 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 368,706 | $ | 17.51 | $ | 1,684,655 | 03/02/10 | 13,578 | 22,630 | 45,260 | (8) | $ | 558,282 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Elliott Sigal, M.D., Ph.D. | $ | 395,850 | $ | 1,218,000 | $ | 3,014,550 | $ | 113,832 | $ | 1,224,000 | $ | 2,988,824 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 3,300 | 14,667 | 32,267 | (3) | $ | 243,032 | 03/02/10 | 2,383 | 14,667 | 24,567 | (4) | $ | 343,354 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,933 | 14,667 | 24,201 | (4) | $ | 229,979 | 03/02/10 | 2,383 | 14,667 | 24,384 | (5) | $ | 343,354 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,933 | 14,667 | 24,201 | (5) | $ | 229,979 | 03/02/10 | 1,634 | 20,103 | 33,673 | (6) | $ | 445,482 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 2,010 | 20,102 | 33,168 | (6) | $ | 298,314 | 03/02/10 | 1,961 | 24,123 | 40,406 | (7) | $ | 596,321 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 24,877 | $ | 435,596 | 03/02/10 | 28,949 | 48,248 | 96,496 | (8) | $ | 1,190,278 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 114,220 | $ | 1,999,992 | $ | 74,400 | $ | 800,000 | $ | 1,953,480 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 295,497 | $ | 17.51 | $ | 1,350,155 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Anthony C. Hooper | $ | 246,997 | $ | 759,992 | $ | 1,880,980 | 03/02/10 | 1,571 | 9,667 | 16,192 | (4) | $ | 226,304 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 1,706 | 7,580 | 16,676 | (3) | $ | 125,601 | 03/02/10 | 1,571 | 9,667 | 16,071 | (5) | $ | 226,304 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 1,933 | 9,667 | 15,951 | (4) | $ | 151,579 | 03/02/10 | 1,359 | 16,722 | 28,009 | (6) | $ | 370,560 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 1,933 | 9,667 | 15,951 | (5) | $ | 151,579 | 03/02/10 | 1,450 | 17,841 | 29,884 | (7) | $ | 441,030 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 1,672 | 16,722 | 27,591 | (6) | $ | 248,154 | 03/02/10 | 21,410 | 35,684 | 71,368 | (8) | $ | 880,324 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 18,778 | $ | 328,803 | 05/03/10 | 20,113 | (9) | $ | 505,440 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Sandra Leung | $ | 50,220 | $ | 540,000 | $ | 1,318,599 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/03/09 | 223,724 | $ | 17.51 | $ | 1,022,217 | 03/02/10 | 1,571 | 9,667 | 16,192 | (4) | $ | 226,304 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/02/10 | 1,571 | 9,667 | 16,071 | (5) | $ | 226,304 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/02/10 | 1,035 | 12,728 | 21,319 | (6) | $ | 282,052 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/02/10 | 1,196 | 14,709 | 24,638 | (7) | $ | 363,606 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/02/10 | 17,651 | 29,419 | 58,838 | (8) | $ | 725,767 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
James M. Cornelius | $ | 87,194 | $ | 937,575 | $ | 2,289,418 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/02/10 | 9,208 | 56,667 | 94,917 | (4) | $ | 1,326,574 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/02/10 | 9,208 | 56,667 | 94,209 | (5) | $ | 1,326,574 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 03/02/10 | 6,935 | 85,304 | 142,884 | (6) | $ | 1,890,337 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (1) | The grant date for annual |
| (2) | Target payouts under |
| (3) | Fair value of performance share unit awards is calculated based on the grant-date closing price of $24.72 on 3/02/2010 and $25.27 on 5/4/2010, a probable outcome of a 100% payout, and is discounted for the 2008-2010 and 2009-2011 awards because no dividends are accrued under these awards. Fair value of market share units is calculated based on a Monte-Carlo simulation. Fair value of restricted stock units is based on the number of units and the grant-date closing price of $25.13 on 05/03/2010. These values are consistent with the grant-date estimate of compensation costs to be recognized over the service period, excluding the effect of forfeitures. |
| (4) | Reflects the third tranche of the |
| (5) | Reflects the |
| (6) | Reflects the |
| (7) |
| (7a) | Reflects the first tranche of the additional performance share units granted under the 2010-2012 award to Mr. Andreotti in order to bring his total long-term incentive value to the level deemed appropriate by the BMS Compensation Committee for his new position as CEO, effective May 4, 2010. |
| (8) | Reflects market share unit awards. They vest in equal annual installments over four years from the grant date. They are earned based on BMS stock performance. Each year when the target annual units are due to vest, the vesting-date stock price (a 10-day average closing price) is divided by the grant price (a 10-day average closing price) to derive a payout factor. Then the payout factor is applied to the target annual units vesting on a given date to derive the payout. These market share units accrue dividend equivalents payable in cash. The same payout factor is applied to derive the dividend payout, if any. The payout threshold equals 60% of the annual target award. The payout maximum equals 200% of the annual target award. |
| (8a) | Reflects the additional 2010 market share unit award granted to Mr. Andreotti in order to bring his total long-term incentive value to the level deemed appropriate by the BMS Compensation Committee for his new position as CEO, effective May 4, 2010. |
| (9) | This restricted stock unit |
Outstanding Equity Awards Atat Fiscal Year-End
20092010 Fiscal Year
| Option Awards | Stock Awards | Option Awards | Stock Awards | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number of Securities Under - - lying Unexer- cised Options(#) | In-the-Money Amount of Unexercised Stock Options ($)(1) | Option Exercise Price | Option Expiration Date | Number of Shares or Units of Stock That Have Not Vested (#)(2) | Market Value of Shares or Units of Stock That Have Not Vested ($) (3) | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Rights That Have Not Vested(#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Rights That Have Not Vested ($) | Number of Securities Under lying Unexercised Options(#) | Option Exercise Price | Option Expiration Date | Number of Shares or Units of Stock That Have Not Vested (#) (2) | Market Value of Shared or Units of Stock That Have Not Vested ($) (3) | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Rights That Have Not Vested ($) (3) | ||||||||||||||||||||||||||||||||||||||||||||
Name | Exer- cisable | Unexer- cisable (1) | Exer- cisable | Unexer- cisable | Exer- cisable | Unexer- cisable(1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||
James M. Cornelius | 360,000 | 0 | (4)(15) | $ | 176,400 | $ | 0 | $ | 24.76 | 10/30/2016 | 95,945 | (9) | $ | 2,422,611 | ||||||||||||||||||||||||||||||||||||||||||||
| 0 | 475,000 | (5)(15) | $ | 0 | $ | 0 | $ | 28.68 | 4/30/2017 | 75,309 | (10) | $ | 1,901,552 | |||||||||||||||||||||||||||||||||||||||||||||
| 1,205,996 | 0 | (6)(15) | $ | 3,750,648 | $ | 0 | $ | 22.14 | 3/3/2018 | 66,612 | (11) | $ | 1,681,953 | |||||||||||||||||||||||||||||||||||||||||||||
| 0 | 1,140,188 | (6)(15) | $ | 0 | $ | 8,825,055 | $ | 17.51 | 3/2/2019 | 86,189 | (12) | $ | 2,176,272 | |||||||||||||||||||||||||||||||||||||||||||||
| 85,142 | (13) | $ | 2,149,836 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 113,607 | (14) | $ | 2,868,577 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1,565,996 | 1,615,188 | $ | 3,927,048 | $ | 8,825,055 | 522,804 | $ | 13,200,801 | ||||||||||||||||||||||||||||||||||||||||||||||||||
Jean-Marc Huet | 72,650 | 0 | (6) | $ | 223,036 | $ | 0 | $ | 22.18 | 3/31/2010 | 0 | $ | 0 | |||||||||||||||||||||||||||||||||||||||||||||
Lamberto Andreotti | 3,365 | 0 | (8) | $ | 0 | $ | 0 | $ | 61.09 | 1/2/2010 | 6,250 | (9) | $ | 157,813 | 36,150 | 0 | (4)(5) | $ | 59.78 | 3/5/2011 | 9,200 | (6) | $ | 243,616 | 39,236 | (7) | $ | 1,038,980 | ||||||||||||||||||||||||||||||
| 26,817 | 0 | (6) | $ | 0 | $ | 0 | $ | 42.64 | 3/6/2010 | 41,667 | (9) | $ | 1,052,092 | 40,000 | 0 | (4) | $ | 48.08 | 3/4/2012 | 13,200 | (6) | $ | 349,536 | 33,575 | (7) | $ | 889,061 | |||||||||||||||||||||||||||||||
| 36,150 | 0 | (6)(16) | $ | 0 | $ | 0 | $ | 59.78 | 3/5/2011 | 18,400 | (9) | $ | 464,600 | 135,000 | 0 | (4) | $ | 23.14 | 3/4/2013 | 22,358 | (6) | $ | 592,040 | |||||||||||||||||||||||||||||||||||
| 40,000 | 0 | (6) | $ | 0 | $ | 0 | $ | 48.08 | 3/4/2012 | 19,800 | (9) | $ | 499,950 | 108,000 | 0 | (4) | $ | 28.11 | 3/1/2014 | 31,032 | (6) | $ | 821,727 | |||||||||||||||||||||||||||||||||||
| 135,000 | 0 | (6) | $ | 284,850 | $ | 0 | $ | 23.14 | 3/4/2013 | 22,358 | (9) | $ | 564,540 | 112,500 | 0 | (4)(8) | $ | 25.45 | 2/28/2015 | 34,986 | (10) | $ | 926,429 | |||||||||||||||||||||||||||||||||||
| 108,000 | 0 | (6) | $ | 0 | $ | 0 | $ | 28.11 | 3/1/2014 | 31,032 | (9) | $ | 783,558 | 115,000 | 0 | (4)(8) | $ | 22.73 | 3/6/2016 | 31,404 | (11) | $ | 831,578 | |||||||||||||||||||||||||||||||||||
| 112,500 | 0 | (6)(15) | $ | 0 | $ | 0 | $ | 25.45 | 2/28/2015 | 19,491 | (10) | $ | 492,148 | 300,000 | 0 | (9)(8) | $ | 24.74 | 11/30/2016 | 72,532 | (12) | $ | 1,920,647 | |||||||||||||||||||||||||||||||||||
| 86,250 | 28,750 | (6)(15) | $ | 217,350 | $ | 72,450 | $ | 22.73 | 3/6/2016 | 17,241 | (11) | $ | 435,335 | 234,720 | 0 | (4)(8) | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||||||||||||||||||||
| 99,999 | 200,001 | (7)(15) | $ | 50,999 | $ | 102,001 | $ | 24.74 | 11/30/2016 | 22,307 | (12) | $ | 563,252 | 305,909 | 0 | (4)(8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||
| 117,360 | 117,360 | (6)(15) | $ | 0 | $ | 0 | $ | 27.01 | 3/5/2017 | 22,037 | (13) | $ | 556,434 | 368,706 | 0 | (4)(8) | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||
| 76,477 | 229,432 | (6)(15) | $ | 237,843 | $ | 713,534 | $ | 22.14 | 3/3/2018 | 34,986 | (14) | $ | 883,397 | 1,755,985 | 0 | 214,712 | $ | 5,685,574 | 72,811 | $ | 1,928,041 | |||||||||||||||||||||||||||||||||||||
| Charles A. Bancroft | 15,775 | 0 | (4)(5) | $ | 59.78 | 3/5/2011 | 1,412 | (13) | $ | 37,390 | 13,578 | (7) | $ | 359,545 | ||||||||||||||||||||||||||||||||||||||||||||
| 18,000 | 0 | (4) | $ | 48.08 | 3/4/2012 | 4,104 | (13) | $ | 108,674 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 26,875 | 0 | (4) | $ | 23.14 | 3/4/2013 | 7,461 | (13) | $ | 197,567 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 20,250 | 0 | (4) | $ | 28.11 | 3/1/2014 | 3,840 | (10) | $ | 101,683 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 16,931 | 0 | (4) | $ | 25.45 | 2/28/2015 | 3,446 | (11) | $ | 91,250 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 19,320 | 0 | (4) | $ | 22.73 | 3/6/2016 | 13,526 | (12) | $ | 358,168 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 16,948 | 5,650 | (4) | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 18,730 | 18,730 | (4) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | 368,706 | (6)(15) | $ | 0 | $ | 2,853,784 | $ | 17.51 | 3/2/2019 | 13,221 | 39,663 | (4) | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||||||
| 841,918 | 944,249 | $ | 791,043 | $ | 3,741,768 | 255,569 | $ | 6,453,117 | 166,050 | 64,043 | 33,789 | $ | 894,733 | 13,578 | $ | 359,545 | ||||||||||||||||||||||||||||||||||||||||||
Elliott Sigal, M.D., Ph.D. | 3,365 | 0 | (8) | $ | 0 | $ | 0 | $ | 61.09 | 1/2/2010 | 5,750 | (9) | $ | 145,188 | 36,150 | 0 | (4)(5) | $ | 59.78 | 3/5/2011 | 7,666 | (6) | $ | 202,996 | 28,949 | (7) | $ | 766,564 | ||||||||||||||||||||||||||||||
| 33,521 | 0 | (6) | $ | 0 | $ | 0 | $ | 42.64 | 3/6/2010 | 16,667 | (9) | $ | 420,842 | |||||||||||||||||||||||||||||||||||||||||||||
| 36,150 | 0 | (6)(16) | $ | 0 | $ | 0 | $ | 59.78 | 3/5/2011 | 15,332 | (9) | $ | 387,133 | |||||||||||||||||||||||||||||||||||||||||||||
| 65,000 | 0 | (6) | $ | 0 | $ | 0 | $ | 48.08 | 3/4/2012 | 20,625 | (9) | $ | 520,781 | 65,000 | 0 | (4) | $ | 48.08 | 3/4/2012 | 13,750 | (6) | $ | 364,100 | |||||||||||||||||||||||||||||||||||
| 68,000 | 0 | (6) | $ | 143,480 | $ | 0 | $ | 23.14 | 3/4/2013 | 22,358 | (9) | $ | 564,540 | 68,000 | 0 | (4) | $ | 23.14 | 3/4/2013 | 22,358 | (6) | $ | 592,040 | |||||||||||||||||||||||||||||||||||
| 48,333 | 0 | (6) | $ | 0 | $ | 0 | $ | 28.11 | 3/1/2014 | 24,877 | (9) | $ | 628,144 | 48,333 | 0 | (4) | $ | 28.11 | 3/1/2014 | 24,877 | (6) | $ | 658,743 | |||||||||||||||||||||||||||||||||||
| 103,500 | 0 | (6)(15) | $ | 0 | $ | 0 | $ | 25.45 | 2/28/2015 | 114,220 | (9) | $ | 2,884,055 | 103,500 | 0 | (4)(8) | $ | 25.45 | 2/28/2015 | 114,220 | (6) | $ | 3,024,546 | |||||||||||||||||||||||||||||||||||
| 71,846 | 23,949 | (6)(15) | $ | 181,052 | $ | 60,351 | $ | 22.73 | 3/6/2016 | 19,491 | (10) | $ | 492,148 | 95,795 | 0 | (4)(8) | $ | 22.73 | 3/6/2016 | 26,772 | (10) | $ | 708,923 | |||||||||||||||||||||||||||||||||||
| 99,999 | 200,001 | (7)(15) | $ | 50,999 | $ | 102,001 | $ | 24.74 | 11/30/2016 | 17,241 | (11) | $ | 435,335 | 199,999 | 100,001 | (9)(8) | $ | 24.74 | 11/30/2016 | 24,031 | (11) | $ | 636,341 | |||||||||||||||||||||||||||||||||||
| 122,250 | 122,250 | (6)(15) | $ | 0 | $ | 0 | $ | 27.01 | 3/5/2017 | 22,307 | (12) | $ | 563,252 | 183,375 | 61,125 | (4)(8) | $ | 27.01 | 3/5/2017 | 28,837 | (12) | $ | 763,604 | |||||||||||||||||||||||||||||||||||
| 76,477 | 229,432 | (6)(15) | $ | 237,843 | $ | 713,534 | $ | 22.14 | 3/3/2018 | 22,037 | (13) | $ | 556,434 | 152,954 | 152,955 | (4)(8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||
| 0 | 295,497 | (6)(15) | $ | 0 | $ | 2,287,147 | $ | 17.51 | 3/2/2019 | 26,772 | (14) | $ | 675,993 | 73,874 | 221,623 | (4)(8) | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||
| 728,441 | 871,129 | $ | 613,375 | $ | 3,163,032 | 327,677 | $ | 8,273,844 | 1,026,980 | 535,704 | 262,511 | $ | 6,951,291 | 28,949 | $ | 766,564 | ||||||||||||||||||||||||||||||||||||||||||
Anthony C. Hooper | 2,524 | 0 | (8) | $ | 0 | $ | 0 | $ | 61.09 | 1/2/2010 | 3,102 | (9) | $ | 78,326 | 36,150 | 0 | (4)(5) | $ | 59.78 | 3/5/2011 | 6,300 | (6) | $ | 166,824 | 21,410 | (7) | $ | 566,947 | ||||||||||||||||||||||||||||||
| 33,127 | 0 | (6) | $ | 0 | $ | 0 | $ | 42.64 | 3/6/2010 | 8,334 | (9) | $ | 210,434 | 40,000 | 0 | (4) | $ | 48.08 | 3/4/2012 | 17,434 | (6) | $ | 461,652 | |||||||||||||||||||||||||||||||||||
| 36,150 | 0 | (6)(16) | $ | 0 | $ | 0 | $ | 59.78 | 3/5/2011 | 12,600 | (9) | $ | 318,150 | 68,000 | 0 | (4) | $ | 23.14 | 3/4/2013 | 13,077 | (6) | $ | 346,279 | |||||||||||||||||||||||||||||||||||
| 40,000 | 0 | (6) | $ | 0 | $ | 0 | $ | 48.08 | 3/4/2012 | 26,151 | (9) | $ | 660,313 | 63,333 | 0 | (4) | $ | 28.11 | 3/1/2014 | 18,778 | (6) | $ | 497,241 | |||||||||||||||||||||||||||||||||||
| 68,000 | 0 | (6) | $ | 143,480 | $ | 0 | $ | 23.14 | 3/4/2013 | 13,077 | (9) | $ | 330,194 | 55,833 | 0 | (4)(8) | $ | 25.45 | 2/28/2015 | 20,113 | (6) | $ | 532,592 | |||||||||||||||||||||||||||||||||||
| 63,333 | 0 | (6) | $ | 0 | $ | 0 | $ | 28.11 | 3/1/2014 | 18,778 | (9) | $ | 474,145 | 78,750 | 0 | (4)(8) | $ | 22.73 | 3/6/2016 | 22,270 | (10) | $ | 589,710 | |||||||||||||||||||||||||||||||||||
| 55,833 | 0 | (6)(15) | $ | 0 | $ | 0 | $ | 25.45 | 2/28/2015 | 12,846 | (10) | $ | 324,362 | 133,333 | 66,667 | (9)(8) | $ | 24.74 | 11/30/2016 | 19,989 | (11) | $ | 529,309 | |||||||||||||||||||||||||||||||||||
| 59,062 | 19,688 | (6)(15) | $ | 148,836 | $ | 49,614 | $ | 22.73 | 3/6/2016 | 11,364 | (11) | $ | 286,941 | 78,462 | 26,154 | (4)(8) | $ | 27.01 | 3/5/2017 | 21,327 | (12) | $ | 564,739 | |||||||||||||||||||||||||||||||||||
| 66,666 | 133,334 | (7)(15) | $ | 34,000 | $ | 68,000 | $ | 24.74 | 11/30/2016 | 14,702 | (12) | $ | 371,226 | 85,747 | 85,748 | (4)(8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||
| 52,308 | 52,308 | (6)(15) | $ | 0 | $ | 0 | $ | 27.01 | 3/5/2017 | 14,525 | (13) | $ | 366,756 | 55,931 | 167,793 | (4)(8) | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||
| 42,873 | 128,622 | (6)(15) | $ | 133,335 | $ | 400,014 | $ | 22.14 | 3/3/2018 | 22,270 | (14) | $ | 562,318 | 695,539 | 346,362 | 139,288 | $ | 3,688,346 | 21,410 | $ | 566,947 | |||||||||||||||||||||||||||||||||||||
| Sandra Leung | 11,831 | 0 | (4)(5) | $ | 59.78 | 3/5/2011 | 6,800 | (6) | $ | 180,064 | 17,651 | (7) | $ | 467,409 | ||||||||||||||||||||||||||||||||||||||||||||
| 0 | 223,724 | (6)(15) | $ | 0 | $ | 1,731,624 | $ | 17.51 | 3/2/2019 | 14,000 | 0 | (4) | $ | 48.08 | 3/4/2012 | 11,940 | (6) | $ | 316,171 | |||||||||||||||||||||||||||||||||||||||
| 519,876 | 557,676 | $ | 459,651 | $ | 2,249,252 | 157,749 | $ | 3,983,162 | 19,310 | 0 | (4) | $ | 23.14 | 3/4/2013 | 14,378 | (6) | $ | 380,729 | ||||||||||||||||||||||||||||||||||||||||
| 15,000 | 0 | (4) | $ | 28.11 | 3/1/2014 | 16,950 | (10) | $ | 448,836 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 15,000 | 0 | (4) | $ | 25.45 | 2/28/2015 | 15,215 | (11) | $ | 402,893 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 14,560 | 0 | (4) | $ | 22.73 | 3/6/2016 | 17,583 | (12) | $ | 465,598 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 66,666 | 33,334 | (9)(8) | $ | 24.74 | 11/30/2016 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 87,075 | 29,025 | (4)(8) | $ | 27.01 | 3/5/2017 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 78,291 | 78,291 | (4)(8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 42,473 | 127,420 | (4)(8) | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 364,206 | 268,070 | 82,866 | $ | 2,194,292 | 17,651 | $ | 467,409 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| James M. Cornelius | 360,000 | 0 | (14)(8) | $ | 24.76 | 10/30/2016 | 113,607 | (10) | $ | 3,008,313 | ||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | 475,000 | (15)(8) | $ | 28.68 | 4/30/2017 | 42,489 | (11) | $ | 1,125,109 | |||||||||||||||||||||||||||||||||||||||||||||||||
| 1,205,996 | 0 | (4)(8) | $ | 22.14 | 3/3/2018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1,140,188 | 0 | (4)(8) | $ | 17.51 | 3/2/2019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2,706,184 | 475,000 | 156,096 | $ | 4,133,422 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (1) | For annual stock option grants in |
| (2) | Represents unvested restricted stock/restricted stock units and annual tranches of the outstanding |
| (3) | Values based on closing stock price on 12/31/ |
| (4) |
| These stock option awards granted on |
| Reflects |
| 100% of award is subject to a 15% price appreciation threshold. Options are not exercisable until the threshold is met and maintained for at least 7 consecutive trading days. The threshold has not been attained for the 5/1/2007 award only. |
| Stock options granted on 12/01/2006 vest in three equal installments at the end of years 3, 4 and 5 following the date of grant. |
| (10) | Reflects the 1st tranche of the 2009-2011performance share unit award at actual performance. |
| (11) | Reflects the 2nd tranche of the 2009-2011 performance share unit award at actual performance. |
| (12) | Reflects the 1st tranche of the 2010-2012 performance share unit award at actual performance. |
| (13) | These restricted stock units granted on 03/06/2007, 03/04/2008, 3/3/2009 vest in equal installments over 4 years following the grant date. |
| (14) | This stock option award granted on 11/01/2006 vested 50% on 03/31/2007. The remaining 50% vested in equal monthly installments over the period of 04/01/2007 through 09/30/2007. |
| (15) | Stock option award granted on 5/1/2007 to Mr. Cornelius upon assuming the role of Chief Executive Officer. The award vested fully at the end of one year following the grant date given that Mr. Cornelius was over age 60 at the time, but it is |
Option Exercises and Stock Vesting
20092010 Fiscal Year
| Options(1) | Stock Awards | Options(1) | Stock Awards | |||||||||||||||||||||||
Name | Number of Shares Acquired on Exercise (#) | Value Realized On Exercise ($) | Number of Shares Acquired on Vesting (#) | Value Realized On Vesting ($) | Number of Shares Acquired on Exercise (#) | Value Realized On Exercise ($) | Number of Shares Acquired on Vesting (#) | Value Realized On Vesting ($) | ||||||||||||||||||
James M. Cornelius | 0 | $ | 0 | 83,844 | $ | 1,550,276(2) | ||||||||||||||||||||
Lamberto Andreotti | 0 | $ | 0 | 63,717 | $ | 1,701,002(2) | ||||||||||||||||||||
| 278,347 | $ | 7,028,262(3) | 116,000 | $ | 3,071,680(3) | |||||||||||||||||||||
Jean-Marc Huet | 0 | $ | 0 | 0 | $ | 0(2) | ||||||||||||||||||||
| 0 | $ | 0(3) | ||||||||||||||||||||||||
Lamberto Andreotti | 0 | $ | 0 | 63,117 | $ | 1,328,663(2) | ||||||||||||||||||||
Charles A. Bancroft | 0 | $ | 0 | 7,747 | $ | 190,521(2) | ||||||||||||||||||||
| 72,043 | $ | 1,819,086(3) | 16,636 | $ | 440,521(3) | |||||||||||||||||||||
Elliott Sigal, M.D., Ph.D. | 0 | $ | 0 | 32,771 | $ | 669,556(2) | 0 | $ | 0 | 36,958 | $ | 962,495(2) | ||||||||||||||
| 72,043 | $ | 1,819,086(3) | 116,000 | $ | 3,071,680(3) | |||||||||||||||||||||
Anthony C. Hooper | 0 | $ | 0 | 21,254 | $ | 422,799(2) | 0 | $ | 0 | 26,453 | $ | 677,403(2) | ||||||||||||||
| 37,233 | $ | 940,133(3) | 76,455 | $ | 2,024,528(3) | |||||||||||||||||||||
Sandra Leung | 0 | $ | 0 | 6,834 | $ | 170,500(2) | ||||||||||||||||||||
| 76,455 | $ | 2,024,528(3) | ||||||||||||||||||||||||
James M. Cornelius | 0 | $ | 0 | 95,945 | $ | 2,361,206(2) | ||||||||||||||||||||
| 369,472 | $ | 9,783,619(3) | ||||||||||||||||||||||||
| (1) | Reflects shares acquired on stock option exercises. None of the executives exercised option awards. |
| (2) | Reflects restricted |
| (3) | Reflects |
Retirement Plan
The following description was effective throughAs of December 31, 2009.2009, we discontinued service accruals under the Retirement program changes were implemented for 2010 which are describedIncome Plan and Benefit Equalization Plan—Retirement Plan in the “Program ChangesU.S. and Puerto Rico for 2010” section above.active plan participants and we stopped adding new participants to those plans. For active plan participants at year-end 2009, we are allowing five additional years of pay growth in the pension plans.
The Retirement Income Plan is a defined benefit pension plan that provides income for employees after retirement. The Retirement Income Plan is a tax-qualified plan, as defined under Section 401(a) of the Internal Revenue Code. The benefit is calculated based on the employee’s final average compensation and years of service. All U.S. employees hired before January 1, 2010 who are not participants in a pension plan through a collective bargaining agreement are eligible for the Retirement Income Plan if they work at least 1,000 hours per year. Employees who participate in the Performance Incentive Plan or whose pay or benefits exceed the IRS qualified plan limits are eligible for the Benefit Equalization Plan—Retirement Plan.
The key plan provisions of the Retirement Income Plan and the Benefit Equalization Plan—Retirement Plan are as follows:
The retirement benefit equals:
2% x Final Average Compensation x Years of Service through December 31, 2009, up to 40, minus
1/70th of the Primary Social Security Benefit x Years of Service through December 31, 2009, up to 40. |
Final Average Compensation equals the average of the five consecutive years out of the last ten years, ending December 31, 2014, in which the employee’s compensation was the highest. Compensation equals the base salary rate, plus bonuses paid during the year. Compensation is subject to the limits defined under Section 401(a)(17) of the Internal Revenue Code.
Normal retirement age is 65. Employees are eligible for early retirement at age 55 with 10 or more years of service.
Employees eligible for early retirement may receive their pension without any reduction at age 60. The pension is reduced by 4% for each year that the retirement age precedes age 60.
Employees are 100% vested after attaining five years of service.
The pension is payable as a monthly life annuity, with or without survivor benefits, or a lump sum.
The Benefit Equalization Plan—Retirement Plan is a non-qualified deferred compensation plan that provides income for employees after retirement in excess of the benefits payable under the qualified Retirement Income Plan. The benefit is calculated using the same formula as the Retirement Income Plan, but without the limits on compensation and benefits imposed under Section 401(a)(17) and Section 415(b) of the Internal Revenue Code. Employees who participate in the Performance Incentive Plan or whose pay or benefits exceed the IRS qualified plan limits are eligible for the Benefit Equalization Plan—Retirement Plan.
The provisions are the same as those above for the Retirement Income Plan, except for the following:
Compensation is not subject to the limits under Section 401(a)(17) of the Internal Revenue Code.
Compensation includes the higher of bonus earned or paid during the year.
The pension is paid as a cash lump sum or, if an election is made at least 12 months prior to retirement, the lump sum may be credited to the Benefit Equalization Plan—Savings Plan. A distribution for an executive classified as a “Specified Employee” of the company, as defined under Section 409A(a)(2)(B)(i) of the Internal Revenue Code, is subject to 409A regulations and is therefore subject to a six-month deferral following the executive’s separation from service.
Present Value of Accumulated Pension Benefits
20092010 Fiscal Year
Name | Plan Name | # of Years of Credited Service | Present Value of Accumulated Benefits (1) | ||||
James M. Cornelius(3) | Retirement Income Plan | 3.7 | $ | 171,098 | |||
| Benefits Equalization Plan | 3.7 | $ | 3,468,430 | ||||
Jean-Marc Huet(4) | Retirement Income Plan | 2.0 | $ | 37,105 | |||
| Benefits Equalization Plan | 2.0 | $ | 385,328 | ||||
Lamberto Andreotti(2) (3) | Retirement Income Plan | 4.3 | $ | 212,307 | |||
| Benefits Equalization Plan | 4.3 | $ | 2,888,209 | ||||
Elliott Sigal, M.D., Ph.D.(3) | Retirement Income Plan | 12.4 | $ | 568,633 | |||
| Benefits Equalization Plan | 12.4 | $ | 5,728,044 | ||||
Anthony C. Hooper(3) | Retirement Income Plan | 14.0 | $ | 545,735 | |||
| Benefits Equalization Plan | 14.0 | $ | 3,164,647 | ||||
Name | Plan Name | # of Years of Credited Service | Present Value of Accumulated Benefits(1) | |||||||
Lamberto Andreotti (2) (3) | Retirement Income Plan | 4.3 | $ | 235,486 | ||||||
| Benefit Equalization Plan | 4.3 | $ | 3,728,396 | |||||||
Charles A. Bancroft | Retirement Income Plan | 25.6 | $ | 890,242 | ||||||
| Benefit Equalization Plan | 25.6 | $ | 2,687,586 | |||||||
Elliott Sigal, M.D., Ph.D. (3) | Retirement Income Plan | 12.4 | $ | 654,629 | ||||||
| Benefit Equalization Plan | 12.4 | $ | 7,583,607 | |||||||
Anthony C. Hooper (3) | Retirement Income Plan | 14.0 | $ | 637,109 | ||||||
| Benefits Equalization Plan | 14.0 | $ | 4,227,716 | |||||||
Sandra Leung | Retirement Income Plan | 17.8 | $ | 627,863 | ||||||
| Benefit Equalization Plan | 17.8 | $ | 2,943,498 | |||||||
James M. Cornelius (4) | Retirement Income Plan | 3.7 | $ | — | ||||||
| Benefit Equalization Plan | 3.7 | $ | — | |||||||
| (1) | The present value of accumulated |
65% lump sumlump-sum utilization for the Retirement Income Plan and 100% lump sumlump-sum utilization for the BenefitsBenefit Equalization Plan
5.75%5.25% discount rate for annuities and 5.75%5.25% discount rate for lump sums
the RP 2000RP-2000 mortality table projected to 20172018 for annuities
the 2010 lump sum2011 lump-sum mortality table under IRC Section 417(e)(3) (combined annuitant and nonannuitant RP2000RP-2000 mortality table with projections blended 50% male/50% female) for lump sums.
These assumptions are the same as those disclosed in conformity with Generally Accepted Accounting Principles. For active executives, payments are assumed to begin at age 60, the earliest age that employees are eligible for an unreduced pension. The actual benefit received will vary based on a number of factors including final pay, years of service and interest rates at the time of retirement. Except for Mr. Cornelius, no pension payments were made to any Named Executive Officer under these plans in 2010.
| (2) | Does not include the value of participation in Italian government pension system. This government-mandated pension system required company contributions to the Italian government while Mr. Andreotti was in Italy and it is legally obliged to provide a pension benefit to Mr. Andreotti. |
| (3) | Mr. Andreotti, Dr. Sigal, and Mr. Hooper have met the requirements for early retirement. |
| (4) |
the 2010 lump-sum RP-2000 mortality table under IRC Section 417(e)(3)
discount rates of 3.26%, 5.24%, and 5.76%, the rates in effect under the three-rate system for distributions from the plan, effective 6/1/2010, the first of the month following his date of separation from service.
Non-Qualified Deferred Compensation Plan
The following description was effective through December 31, 2009. Plan changes were implemented for 2010 which are described in the “Program Changes for 2010” section above.
The Benefit Equalization Plan—Savings Plan is a non-qualified deferred compensation plan that allows employees to defer a portion of their base salarytotal cash compensation and to receive company matching contributions in excess of contributions allowed under the Savings and Investment Program. The Savings and Investment Program is a tax-qualified plan, as defined under Section 401(a) and Section 401(k) of the Internal Revenue Code. Employees who are eligible to participate in the Performance Incentive Plan orBristol-Myers Squibb Savings and Investment Program, and whose pay or benefits exceed the IRS qualified plan limits, are eligible for the Benefit Equalization Plan—Savings Plan. The key provisions of the BEP-Savings Plan, which was enhanced in the U.S. and Puerto Rico as of January 1, 2010, are as follows:
Employee deferrals to the BEP-Savings Plan begin once the employee’s total base salaryeligible compensation paid for the year exceeds the limit under Section 401(a)(17) of the Internal Revenue Code, or total contributions to the Savings and Investment Program exceed the limits under Section 415(c) of the Internal Revenue Code.
Employees may defer up to 20% of their base salary.eligible compensation.
The company matching contribution equals 75%100% of the employee’s contribution up toon the first 6% of base salary.eligible compensation that an employee elects to contribute.
An additional automatic company contribution, which is based on a point system of one’s age plus service, equals: below 40 points—3% of total cash; between 40 and 60 points—4.5%; and at 60 points and above—6%. For those employees with 60 or more points who had 10 or more years of service at year-end 2009, an additional automatic contribution of 2% is provided for a five-year period.
The plan is not funded. Benefits are paid from general assets of the company.
Employees may allocate their contributions among 13 different investment options.
The employee’s full balance under the BEP-Savings Plan is paid following termination of employment, or, if eligible, an election iscan be made at least 12 months prior to termination, theseparation from service to defer payments may be deferred until a later date, no sooner than five years following the termination date.date of
separation from service. A distribution for an executive classified as a “Specified Employee” of the company, as defined under Section 409A(a)(2)(B)(i) of the Internal Revenue Code, is subject to 409A regulations and is therefore subject to a six-month deferral following the executive’s separation from service. |
Non-Qualified Deferred Compensation
20092010 Fiscal Year
Name | Executive Contributions in Last FY | Registrant Contributions in Last FY | Aggregate Earnings in Last FY(3) | Aggregate Withdrawals/ Distributions | Aggregate Balance at Last FYE(4) | ||||||||||
James M. Cornelius (1) | $ | 150,600 | $ | 56,475 | $ | 152,137 | $ | 0 | $ | 631,602 | |||||
Jean-Marc Huet (1) (5) | $ | 66,836 | $ | 30,076 | $ | 20,486 | $ | 0 | $ | 157,442 | |||||
Lamberto Andreotti (1) | $ | 59,984 | $ | 44,988 | $ | 28,816 | $ | 0 | $ | 360,687 | |||||
Elliott Sigal M.D., Ph.D. (1) | $ | 92,382 | $ | 34,643 | $ | 42,381 | $ | 0 | $ | 854,885 | |||||
Elliott Sigal M.D., Ph.D. (2) | $ | 0 | $ | 0 | $ | 82,564 | $ | 0 | $ | 1,428,967 | |||||
Anthony C. Hooper (1) | $ | 80,596 | $ | 24,179 | $ | 99,975 | $ | 0 | $ | 815,476 | |||||
Name | Executive Contributions in Last FY | Registrant Contributions in Last FY(1) | Aggregate Earnings in Last FY(2) | Aggregate Withdrawals/ Distributions | Aggregate Balance at Last FYE (1) (3) | |||||||||||||||
Lamberto Andreotti(4) | $ | 250,756 | $ | 585,098 | $ | 75,626 | $ | 0 | $ | 1,272,167 | ||||||||||
Charles A. Bancroft(4) | $ | 46,333 | $ | 113,009 | $ | 21,074 | $ | 0 | $ | 263,157 | ||||||||||
Elliott Sigal M.D., Ph.D.(4) | $ | 355,785 | $ | 417,533 | $ | 54,545 | $ | 0 | $ | 1,682,750 | ||||||||||
Elliott Sigal M.D., Ph.D.(5) | $ | 0 | $ | 0 | $ | 52,971 | $ | 0 | $ | 1,481,938 | ||||||||||
Anthony C. Hooper(4) | $ | 252,006 | $ | 253,872 | $ | 113,527 | $ | 0 | $ | 1,434,881 | ||||||||||
Sandra Leung(4) | $ | 202,141 | $ | 208,265 | $ | 71,529 | $ | 0 | $ | 707,746 | ||||||||||
James M. Cornelius(4) (6) | $ | 572,979 | $ | 582,779 | $ | 229,047 | $ | 2,016,407 | $ | 0 | ||||||||||
| (1) | Includes the additional annual registrant contributions earned in 2010 but allocated in February 2011. |
| (2) | The company does not pay above-market interest rates on non-qualified deferred compensation. |
| (3) | Portions of the amounts shown in this column have been included in 2010 compensation reflected in the Salary and All Other Compensation columns of the Summary Compensation Table, as follows: |
| 2010 | ||||||||||||
Name | Salary | All Other Compensation | Total | |||||||||
Lamberto Andreotti | $ | 250,756 | $ | 585,098 | $ | 835,854 | ||||||
Charles A. Bancroft | $ | 46,333 | $ | 113,009 | $ | 159,342 | ||||||
Elliott Sigal M.D., Ph.D. | $ | 355,785 | $ | 417,533 | $ | 773,319 | ||||||
Anthony C. Hooper | $ | 252,006 | $ | 253,872 | $ | 505,878 | ||||||
Sandra Leung | $ | 202,141 | $ | 208,265 | $ | 410,406 | ||||||
James M. Cornelius | $ | 572,979 | $ | 582,779 | $ | 1,155,758 | ||||||
In addition, portions of the aggregate balances in this column reflect amounts from 2006 through 2009, which were reported in the last four Proxy Statements, and amounts from earlier years, which were reported in accordance with the then applicable proxy disclosure rules.
| (4) | Reflects non-qualified savings and investment program. Executive contributions are included in the Salary column and registrant contributions are included in the All Other Compensation column of the Summary Compensation Table. |
| Reflects earnings and aggregate balance related to prior voluntary deferral of |
| 2009 | |||||||||
Name | Salary | All Other Compensation | Total | ||||||
James M. Cornelius | $ | 150,600 | $ | 56,475 | $ | 207,075 | |||
Jean-Marc Huet | $ | 66,836 | $ | 30,076 | $ | 96,913 | |||
Lamberto Andreotti | $ | 59,984 | $ | 44,988 | $ | 104,972 | |||
Elliott Sigal M.D., Ph.D. | $ | 92,382 | $ | 34,643 | $ | 127,024 | |||
Anthony C. Hooper | $ | 80,596 | $ | 24,179 | $ | 104,775 | |||
Portions of the aggregate balances in this column reflect amounts from 2006, 2007, and 2008, which were reported in the last three Proxy Statements, and amounts from earlier years, which were reported in accordance with the then applicable proxy disclosure rules.
The following illustrates the potential payments and benefits under the company’s plans and programs to the Named Executive Officers upon a termination of employment assuming an effective date of December 31, 2009.2010. To the extent payments and benefits are generally available to salaried employees on a non-discriminatory basis, they are excluded from the table.
Termination of Employment Obligations (Excluding Vested Benefits)
20092010 Fiscal Year
Name | Cash Severance (1) | Non-Equity Annual Incentive (2) | In the Money Value of Options (3) | Restricted Stock (4) | Long-Term Performance Plan (5) | Retirement (6) | Savings Plan Balances (7) | Health (8) | Retiree Medical (9) | Other | Total | Gross-Up on Excise Taxes (10) | |||||||||||||||||||||||||
Involuntary Termination Not for Cause | |||||||||||||||||||||||||||||||||||||
James M. Cornelius(11) | $ | 3,000,000 | $ | 4,494,825 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 37,192 | $ | 0 | $ | 0 | $ | 7,532,017 | $ | 0 | |||||||||||||
Lamberto Andreotti(11) | $ | 8,790,510 | $ | 3,076,402 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 25,543 | $ | 0 | $ | 0 | $ | 11,892,455 | $ | 0 | |||||||||||||
Elliott Sigal, M.D., Ph.D.(11) | $ | 2,040,000 | $ | 2,189,879 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 23,466 | $ | 0 | $ | 0 | $ | 4,253,345 | $ | 0 | |||||||||||||
Anthony C. Hooper(11) | $ | 1,600,000 | $ | 1,113,373 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 21,983 | $ | 0 | $ | 0 | $ | 2,735,356 | $ | 0 | |||||||||||||
Qualifying Termination Within 3 Years Following a Change in Control | |||||||||||||||||||||||||||||||||||||
James M. Cornelius(12) | $ | 11,212,500 | $ | 4,494,825 | $ | 8,825,055 | $ | 2,422,611 | $ | 0 | $ | 3,450,215 | $ | 0 | $ | 114,364 | $ | 944 | $ | 0 | $ | 30,520,515 | $ | 5,995,543 | |||||||||||||
Lamberto Andreotti(12) | $ | 8,790,510 | $ | 3,076,402 | $ | 2,853,784 | $ | 2,637,236 | $ | 0 | $ | 3,091,961 | $ | 0 | $ | 79,937 | $ | 0 | $ | 0 | $ | 20,529,830 | $ | 5,165,939 | |||||||||||||
Elliott Sigal, M.D., Ph.D.(12) | $ | 6,709,560 | $ | 2,189,879 | $ | 2,287,147 | $ | 4,877,366 | $ | 0 | $ | 3,663,525 | $ | 0 | $ | 73,743 | $ | 0 | $ | 0 | $ | 19,801,219 | $ | 5,699,341 | |||||||||||||
Anthony C. Hooper(12) | $ | 4,784,000 | $ | 1,113,373 | $ | 1,731,624 | $ | 1,544,467 | $ | 0 | $ | 2,907,324 | $ | 0 | $ | 69,315 | $ | 0 | $ | 0 | $ | 12,150,103 | $ | 3,642,519 | |||||||||||||
Voluntary Termination | |||||||||||||||||||||||||||||||||||||
Jean-Marc Huet(13) | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 104,156 | (14) | $ | 104,156 | $ | 0 | ||||||||||||
Name | Cash Severance (1) | Non-Equity Annual Incentive (2) | In the Money Value of Options (3) | Restricted Stock/ Restricted Stock Units (4) | Market Share Units (5) | Perfor- mance Share Units (6) | Retirement (7) | Savings Plan Balances (8) | Health (9) | Retiree Medical (10) | Total | Gross-Up on Excise Taxes (11) | ||||||||||||||||||||||||||||||||||||
Involuntary Termination Not for Cause |
| |||||||||||||||||||||||||||||||||||||||||||||||
Lamberto Andreotti(12) | $ | 2,800,000 | $ | 3,131,544 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 13,963 | $ | 0 | $ | 5,945,507 | $ | 0 | ||||||||||||||||||||||||
Charles A. Bancroft | $ | 1,500,000 | $ | 978,595 | $ | 437,065 | $ | 129,620 | $ | 0 | $ | 551,102 | $ | 1,681,984 | $ | 0 | $ | 20,567 | $ | 117,310 | $ | 5,416,243 | $ | 0 | ||||||||||||||||||||||||
Elliott Sigal, M.D., Ph.D.(12) | $ | 2,040,000 | $ | 1,952,642 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 23,708 | $ | 0 | $ | 4,016,350 | $ | 0 | ||||||||||||||||||||||||
Anthony C. Hooper(12) | $ | 1,600,000 | $ | 1,276,236 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 21,149 | $ | 0 | $ | 2,897,385 | $ | 0 | ||||||||||||||||||||||||
Sandra Leung | $ | 1,350,000 | $ | 797,648 | $ | 1,540,742 | $ | 249,971 | $ | 0 | $ | 1,317,327 | $ | 1,672,243 | $ | 0 | $ | 20,341 | $ | 92,371 | $ | 7,040,643 | $ | 0 | ||||||||||||||||||||||||
Qualifying Termination Within 3 Years Following a Change in Control |
| |||||||||||||||||||||||||||||||||||||||||||||||
Lamberto Andreotti(13) | $ | 10,465,000 | $ | 3,131,544 | $ | 0 | $ | 1,313,302 | $ | 3,439,355 | $ | 0 | $ | 3,350,530 | $ | 0 | $ | 42,706 | $ | 0 | $ | 21,742,437 | $ | 5,801,359 | ||||||||||||||||||||||||
Charles A. Bancroft | $ | 4,485,000 | $ | 978,595 | $ | 437,065 | $ | 343,631 | $ | 643,994 | $ | 551,102 | $ | 6,885,393 | $ | 0 | $ | 64,542 | $ | 114,192 | $ | 14,503,514 | $ | 5,668,418 | ||||||||||||||||||||||||
Elliott Sigal, M.D., Ph.D.(13) | $ | 6,709,560 | $ | 1,952,642 | $ | 0 | $ | 3,595,587 | $ | 1,373,041 | $ | 0 | $ | 2,327,521 | $ | 0 | $ | 73,915 | $ | 0 | $ | 16,032,266 | $ | 4,009,911 | ||||||||||||||||||||||||
Anthony C. Hooper(13) | $ | 4,784,000 | $ | 1,276,236 | $ | 0 | $ | 1,470,567 | $ | 1,015,508 | $ | 0 | $ | 2,664,784 | $ | 0 | $ | 66,200 | $ | 0 | $ | 11,277,295 | $ | 3,224,443 | ||||||||||||||||||||||||
Sandra Leung | $ | 3,632,850 | $ | 797,648 | $ | 1,540,742 | $ | 876,965 | $ | 837,218 | $ | 1,317,327 | $ | 4,560,659 | $ | 0 | $ | 63,867 | $ | 94,988 | $ | 13,722,264 | $ | 4,608,906 | ||||||||||||||||||||||||
| (1) |
| (2) | Reflects |
| (3) | Intrinsic values as of 12/31/ |
| (4) | Values as of 12/31/ |
| (5) | Values as of 12/31/ |
| (6) | Values as of 12/31/2010 based on the closing stock price of $26.48 on 12/31/2010. For both involuntary termination and change in control, represents actual payouts of the first and second tranches of the |
| Change-in-control values include special early retirement subsidy and additional years of credited service and age. |
| Reflects vesting of unvested portion of employer matching contributions upon change in control as of 12/31/ |
| For involuntary termination, reflects health care continuation through the severance period. For change in control, represents continuation of health benefits for 3 years. |
| Change-in-control values |
| Reflects the gross-up under the change-in-control agreements. The excise tax amount on the excess parachute payment (i.e., the amount subject to the excise tax) is grossed up to account for the effect of federal and state income taxes, and the excise tax. Includes Federal income tax of 35%, excise tax of 20% and relevant state taxes. Does not reflect employment taxes or amounts attributable to the loss of itemized deductions. |
| These |
| These |
Options—The value of the 2009 stock option award which will not have been held for one year from the grant date.
Restricted Stock—Stock/Restricted Stock Units—The difference between a pro-rata portion of restricted stock/restricted stock units held for one year from the grant date and all unvested restricted stock/restricted stock units including sharesshares/units held less than one year.
Market Share Units—The difference between a pro-rata portion of market share units held for one year from the grant date and all unvested market share units including units held less than one year from the grant date, subject to performance provisions. Following is a description of |
We have described below the payments and benefits available under variousdifferent termination scenarios as of December 31, 2009:scenarios:
Voluntary Termination
The company does not offer any payments or benefits to salaried employees, including the Named Executive Officers, upon a voluntary termination other than those that are vested at the time of termination.
Retirement and Death
The following benefits are generally available to all salaried employees including the Named Executive Officers:
Annual Incentive—Employees are eligible for a pro-rata award based on the number of months worked in the performance period.
Stock Options—Employees are eligible for accelerated vesting of stock options held at least one year from the grant date and hashave the full term to exercise. Forexercise vested stock options. Upon retirement, exercise thresholds, as described in the Compensation Discussion and Analysis (CD&A) and Outstanding Equity Awards Table, where applicable, remain in effect.effect, where applicable. Upon death, exercise thresholds lapse.
Restricted Stock/Restricted Stock Units—Employees are eligible for a pro-rata portion of restricted stock/restricted stock unit awards held at least one year from the grant date.
Long-Term Market Share Units—Employees are eligible for a pro-rata portion of market share unit awards held at least one year from the grant date, subject to performance provisions.
Performance Share Unit Awards—Employees are eligible for a pro-rata portion of long-term performance share unit awards held at least one year from the grant date.
Defined Benefit Pension Plans—Employees are eligible for benefits accrued under the Retirement Income Plan and the Benefit Equalization Plan—Retirement Plan.
Savings Plans—Employees are eligible for benefits accumulated under the Savings and Investment Program and the Benefit Equalization Plan—Savings Plan.
Post-Retirement Medical and Life Insurance—Employees with ten years of service are eligible for post-retirement medical and life insurance benefits.
Involuntary Termination Not for Cause
The following benefits are generally available to all salaried employees including the Named Executive Officers:
Annual Incentive—Employees are eligible for a pro-rata award based on the number of months worked in the performance period if the termination occurs on or after September 30th of the plan year. If an employee is eligible to retire, or the employee’s age plus years of service equal or exceed 70, and the employee has at least 10 years of service, the employee is eligible for a pro-rata award based on the number of months worked in the performance period.
Stock Options—Upon signing a general release, an employee is eligible for accelerated vesting of stock options held at least one year from the grant date and has three months to exercise. If an employee is eligible to retire, or the employee’s age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee will have the full term to exercise. Exercise thresholds, as described in the CD&A and Outstanding Equity Awards Table, where applicable, remain in effect.effect, where applicable.
Restricted Stock/Restricted Stock Units—Upon signing a general release, employees are eligible for a pro-rata portion of restricted stock/restricted stock unit awards held at least one year from the grant date.
Long-Term Performance AwardsMarket Share Units—Awards granted in 2007 and earlierUpon signing a general release, employees are forfeited; however, if the employee is eligible to retire or the employee’s age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee will be eligible for a pro-rata portion of anyunvested market share unit awards held at least one year from the grant date. For awards granted in 2008 or later, the employee willdate, subject to performance provisions.
Performance Share Unit Awards—Upon signing a general release, employees are be eligible for a pro-rata portion of any performance share unit awards held at least one year from the grant date.
Defined Benefit Pension Plans—Employees are eligible for benefits accrued under the Retirement Income Plan and the Benefit Equalization Plan—Retirement Plan. If the employee’s age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee is not eligible for early retirement, and the employee signs a general release, the retirement benefits are payable immediately following termination of employment with thebased upon enhanced adjustment factors that are applicablesimilar to those applied to employees eligible for early retirement.
Savings Plans—Employees are eligible for benefits accumulated under the Savings and Investment Program and the Benefit Equalization Plan—Savings Plan.
Post-Retirement Medical Insurance—If the employee’s age plus years of service equal or exceed 70 and the employee has at least 10 years of service, the employee is not eligible for early retirement, and the employee signs a general release, the employee is eligible for continued medical coverage without company subsidy until age 55, and is eligible for thecompany-subsidized, post-retirement medical benefits starting at age 55.
Cash Severance—As disclosed in the CD&A, ourapproximately 14 senior executives (including the Named Executive OfficersOfficers) are eligible to receive severance payments and benefits if their employment is terminated for any of the following reasons:
Involuntary termination not for “cause”;
The executive’s monthly base salary is reduced;
The executive’s grade level is reduced; or
The location of the executive’s job or office is changed, so that it will be based at a location which is more than 50 miles further (determined in accordance with the company’s relocation policy) from their primary residence than their work location immediately prior to the proposed change in their job or office.
A terminated executive who signs a general release will be eligible for the following:
Severance payments in the amount of 2 times base salary for all NEOs, except for Mr. Andreotti who is eligible for 2.99our senior-most executives, including the Named Executive Officers, and 1.5 times base salary and target bonus under a previously disclosed arrangement. The benefit for Mr. Andreotti is based on the benefit that he was entitled to receive as an employee in Italy prior to becoming a local U.S. employee. Upon becoming CEO, Mr. Andreotti will no longer be eligible to receive this special severance arrangement;other senior executives;
Continuation of medical, dental and life insurance benefits; and
Outplacement services.
Change in Control
As disclosed in the CD&A, the company has entered into change-in-control agreements with certain senior executives, including all of the Named Executive Officers. The current agreements will expire on December 31, 2011, and may be extended with revisions, as appropriate, beginning on
January 1, 2012, in one-year increments unless either the company or the executive gives prior notice of termination of the agreement or a change in control shall have occurred prior to January 1 of such year.
To trigger benefits, under these agreements, there must be both a change in control of the company and either (i) a subsequent involuntary termination without cause by the company or (ii) a good reason termination by the employee. Good reason includes a reduction in job responsibilities or changes in pay and benefits as well as relocation beyond 50 miles. The executive has 120 days to assert a claim for payments under this provision. This protection extends for three years following a change in control.
“Change in Control” means the occurrence of any one of the following events after the Effective Date:
| (i) | Any Person (as defined in Section 13(d)(3) of the Securities and Exchange Act) shall have become the direct or indirect beneficial owner of thirty percent (30%) or more of the then outstanding common shares of the company; |
| (ii) | The consummation of a merger or consolidation of the company with any other corporation other than (A) a merger or consolidation which would result in the voting securities of the company outstanding immediately prior thereto continuing to represent at least |
| (iii) | The date the stockholders of the company approve a plan of complete liquidation of the company or an agreement for the sale or disposition by the company of all or substantially all the company’s assets; or |
| (iv) | The date there shall have been a change in the composition of the Board of Directors of the company within a two-year period such that a majority of the Board does not consist of directors who were serving at the beginning of such period together with directors whose initial nomination for election by the company’s stockholders or, if earlier, initial appointment to the Board, was approved by the vote of two-thirds of the directors then still in office who were in office at the beginning of the two-year period together with the directors who were previously so approved. |
A terminated Named Executive Officer who signs an agreement not to compete with the company for a period of one year, shall receive the following benefits:
A cash payment equal to 2.99 years of base salary plus target bonus. (If time between termination date and normal retirement is less than 2.99 years, then payment multiple is equal to this lesser amount of time.)
Payout of outstanding performance share unit awards under the Long-Term Performance Award Program on a pro-rata basis at target including any award held less than one year. Banked portions paid out in full.
Payout of annual bonus on a pro-rata basis at target.
Vesting of unvested market share units, subject to performance provisions, including units held less than one year.
Vesting of unvested stock options, including options held less than one year. Waiver of exercise thresholds placed on awards, where applicable.
Vesting of restricted stock/restricted stock units, including shares/units held less than one year.
Three additional years of service and age for pension purposes.
Application of the early retirement subsidy under the Retirement Income Plan for executives who are terminated prior to age 55 as if the executive were 55 with at least ten years of service.
Eligibility for retiree medical benefits based on three years additional age and service.
Continuation of benefits for three years.
Vesting of unvested match in the Company Savings Plan.
Gross-up of excise tax on payments deemed to be excess parachute payments that exceed 10% of the total payment which could be made without triggering the golden parachute excise taxes under Sections 280G and 4999 of the Internal Revenue Code.
Payment of any legal fees incurred to enforce the agreement.
VOTING SECURITIES AND PRINCIPAL HOLDERS
AtAs mentioned in the close of business on March 11, 2010, there were 1,717,916,857 shares of $0.10 par value common stock and 5,345 shares of $2.00 convertible preferred stock outstanding and entitled to vote.
Common Stock Ownership by Directors and Executive Officers
The following table sets forth, as of February 16, 2010, beneficial ownership of shares of our common stock by each director, each ofCD&A, the named executive officers and all directors and executive officers as a group. Shares are beneficially owned when an individualcompany has voting and/or investment power over the shares or could obtain voting and/or investment power over the shares within 60 days. Voting power includes the power to direct the voting of the shares and investment power includes the power to direct the disposition of the shares. Unless otherwise noted, shares listed below are owned directly or indirectly with sole voting and investment power. None of our directors and executive officers, individually or as a group, beneficially owns greater than 1% of our outstanding shares of common or preferred stock.
Name | Bristol-Myers Squibb Company | |||||
| Total Common Shares Owned(1) | Common Shares Underlying Options or Restricted Stock Units(2) | Common Shares Underlying Deferred Share Units(3) | ||||
L. Andreotti | 1,282,955 | 1,101,236 | 0 | |||
L. B. Campbell | 35,765 | 20,810 | 14,955 | |||
J. M. Cornelius | 3,043,757 | 2,806,504 | 4,936 | |||
L. J. Freeh | 25,421 | 2,500 | 22,921 | |||
L. H. Glimcher, M.D. | 71,847 | 20,810 | 51,037 | |||
M. Grobstein | 26,373 | 0 | 22,990 | |||
A. C. Hooper | 767,624 | 670,716 | 0 | |||
L. Johansson | 56,941 | 20,810 | 34,131 | |||
A. J. Lacy | 17,578 | 0 | 15,273 | |||
V. L. Sato, Ph.D. | 21,054 | 0 | 21,054 | |||
E. Sigal, M.D., Ph.D. | 1,143,485 | 967,376 | 21,652 | |||
T. D. West, Jr. | 16,367 | 0 | 16,367 | |||
R. S. Williams, M.D. | 28,075 | 0 | 27,575 | |||
All Directors and Executive Officers as a Group | 9,330,242 | 8,040,190 | 252,891 | |||
Principal Holders of Voting Securities
The following table sets forth information regarding beneficial owners of more than 5 percent of the outstanding shares of our common stock. There are no beneficial owners of more than 5 percent of the outstanding shares of our preferred stock.
Name | Number of Shares Beneficially Owned | Percent of Class | ||||
BlackRock Inc. 40 East 52nd Street New York, NY 10022 | 135,563,389 | (1) | 7.92 | %(1) |
Section 16(a) Beneficial Ownership Reporting Compliance
Under Section 16(a) of the Securities Exchange Act of 1934, our directors, executive officers and the beneficial holders of more than 10% of our common stock are required to file reports of ownership and changes in ownership with the U.S. Securities and Exchange Commission. To the best of our knowledge, during 2009 all applicable Section 16(a) filing requirements were met, except that, due to administrative errors, Forms 4 were filed late for each director relatedmade certain amendments to the annual grant of deferred share units andchange-in-control agreements for each executive officer relatednewly-eligible executives on or after September 1, 2010. Those amendments, which are described in the CD&A, do not apply to shares that were “banked” but not vested under the long-term performance awards. In addition, a Form 5 was filed for Michael Grobstein disclosing his exchange of 1,617 shares of company common stock for 1,020 shares of common stock of Mead Johnson Nutrition Company in a company exchange offer.Named Executive Officers.
ITEM 2—RATIFICATION OF THE APPOINTMENT OF INDEPENDENT
REGISTERED PUBLIC ACCOUNTING FIRM
Our Board of Directors, upon the recommendation of its Audit Committee, has ratified the Audit Committee’s appointment of Deloitte & Touche LLP as our independent registered public accounting firm for the year 2010.2011. As a matter of good corporate governance, we are asking stockholders to ratify such appointment. In the event our stockholders fail to ratify the appointment, it will be considered as a direction to our Board of Directors and the Audit Committee to appoint another independent registered public accounting firm. It is understood that even if the appointment is ratified, the Audit Committee at its discretion, may direct the appointment of a new independent registered public accounting firm at any time during the year if the Audit Committee feels that such a change would be in the best interests of our company and our stockholders.
Representatives from Deloitte & Touche LLP will be present at the Annual Meeting to respond to appropriate questions and to make any statements as they may desire.
The Board of Directors unanimously recommends a vote “FOR” the ratification of the appointment of Deloitte & Touche LLP as Bristol-Myers Squibb’s independent registered public accounting firm for 2010.2011.
The following table presents aggregate fees for professional audit services rendered by Deloitte & Touche LLP (D&T) for the years ended December 31, 20092010 and 20082009 for the audits of our annual financial statements and internal control over financial reporting, and fees billed for other services rendered by D&T during those periods. The fees include those related to the separate audit in 2009 of Mead Johnson Nutrition Company (MJN), a public subsidiary of the company that was divested through a split-off transaction in December 2009.2009 and certain tax services provided by BMS under a Transition Services Agreement (TSA) with MJN for which Deloitte was engaged to perform in 2010.
| 2008 | 2009(1) | 2009(1) | 2010 | |||||||||||
| (in millions) | (in millions) | |||||||||||||
Audit | $ | 16.73 | $ | 17.27 | $ | 17.27 | $ | 13.03 | ||||||
Audit Related | 7.22 | 3.73 | 3.73 | 1.72 | ||||||||||
Tax | 7.02 | 7.22 | 7.22 | 7.68 | ||||||||||
All Other | .10 | .29 | .29 | .01 | ||||||||||
Total | $ | 31.07 | $ | 28.51 | $ | 28.51 | $ | 22.44 | ||||||
| (1) | Fees for professional audit services provided solely to BMS (excluding fees for services provided to MJN) during 2009 and 2010 were: |
| 2009 BMS | 2009 BMS | 2010 BMS | |||||||||
| (in millions) | (in millions) | (in millions) | |||||||||
Audit | $ | 13.86 | $ | 13.86 | $ | 13.03 | |||||
Audit Related | 2.78 | 2.78 | 1.72 | ||||||||
Tax | 7.18 | 7.18 | 6.65 | ||||||||
All Other | .01 | .01 | .01 | ||||||||
Total | $ | 23.83 | $ | 23.83 | $ | 21.41 | |||||
Audit fees for 20082009 and 20092010 were for professional services rendered for the audits of our consolidated financial statements, including accounting consultation, and of our internal controlscontrol over financial reporting in accordance with Section 404 of the Sarbanes-Oxley Act (Section 404), statutory and subsidiary audits, timely reviews of quarterly financial statements, consents, issuance of comfort letters, and assistance with review of documents filed with the SEC.
Audit Relatedfees for 20082009 and 20092010 were primarily for assurance services, employee benefit plan audits, due diligence related to acquisitions and divestitures, divestiture audits, the MJN partial IPOinitial public offering in 2009 and services that are not required by statute or regulation.
Taxfees for 20082009 and 20092010 were for services related to tax compliance, including the preparation of tax returns and claims for refund, tax planning (excluding planning related to transactions or proposals for which the sole purpose may be tax avoidance or for which tax treatment may not be supported by the Internal Revenue Code) and tax advice, including assistance with and representation in tax audits and appeals, advice related to divestitures and acquisitions, preparation of individual income tax returns (excluding those of executive officers) and consultations relating to our expatriate program and international compensation matters, and requests for rulings or technical advice from tax authorities. For 2010 tax fees also included preparation of tax returns and certain planning activities for which Deloitte was engaged to perform under the TSA for tax services.
All Other fees for 20082009 and 20092010 related to license fees for accounting and reporting research library services, surveys, benchmarking and training programs.
Pre-Approval Policy for Services Provided by our Independent Registered Public Accounting Firm
The Audit Committee has established a policy to pre-approve all audit and permissible non-audit services provided by our independent registered public accounting firm consistent with applicable SEC rules. Our independent registered public accounting firm is generally prohibited from performing any management consulting projects. Our independent registered public accounting firm is also prohibited from providing tax consulting services relating to transactions or proposals in which the sole purpose may be tax avoidance or for which the tax treatment may not be supported by the Internal Revenue
Code. Prior to the engagement of our independent registered public accounting firm for the next year’s audit, management submits an aggregate of services expected to be rendered during that year for each of the four categories of services described above to the Audit Committee for approval. Prior to engagement, the Audit Committee pre-approves these services by category of service. The fees are budgeted by category of service and the Audit Committee receives periodic reports from management and our independent registered public accounting firm on actual fees versus the budget by category of service. During the year, circumstances may arise when it may become necessary to engage our independent registered public accounting firm for additional services not contemplated in the pre-approval. In those instances, the Audit Committee requires specific pre-approval before engaging our independent registered public accounting firm.
The Audit Committee may delegate pre-approval authority to one or more of its members. The member or members to whom such authority is delegated is required to report, for informational purposes, any pre-approval decisions to the Audit Committee at its next regularly scheduled meeting.
Prior to the split-off in December 2009, engagements of Deloitte & Touche LLP by MJN were reviewed and approved by the audit committee of MJN pursuant to pre-approval policies adopted by MJN’s audit committee. For 2010, the MJN audit committee also approved the engagement of Deloitte to perform certain tax services required by the TSA.
As the Audit Committee of the Board of Directors, we are composed of independent directors as required by and in compliance with the listing standards of the New York Stock Exchange. We operate pursuant to a written charter adopted by the Board of Directors that is published on the company’s website.
Management has primary responsibility for the company’s financial reporting process, principles and internal controls as well as preparation of its consolidated financial statements. The independent registered public accounting firm is responsible for performing an audit in accordance with the
standards of the Public Company Accounting Oversight Board (United States) (PCAOB) to obtain reasonable assurance that Bristol-Myers Squibb’s consolidated financial statements are free from material misstatement and expressing an opinion on the conformity of such financial statements with accounting principles generally accepted in the United States. We are responsible for overseeing and monitoring D&T’s auditing process on behalf of the Board of Directors.
As part of the oversight of our financial statements, we review and discuss with both management and D&T all annual and quarterly financial statements prior to their issuance. Management advised us that each set of financial statements reviewed was prepared in accordance with accounting principles generally accepted in the United States. We have reviewed with management significant accounting and disclosure issues and reviewed with D&T matters required to be discussed pursuant to auditing standards adopted by the PCAOB.
In addition, we have received the written disclosures and the letter from D&T required by PCAOB Ethics and Independence Rule 3526, “Communication with Audit Committees Concerning Independence”, and have discussed with D&T their independence from Bristol-Myers Squibb and its management. We have also determined that D&T’s provision of non-audit services in 20092010 was compatible with, and did not impair, its independence. We have also received written materials addressing D&T’s internal quality control procedures and other matters, as required by the New York Stock Exchange listing standards.
We have discussed with our internal auditors and D&T the overall scope and plans for their respective audits. We have met with the internal auditors and D&T, with and without management present, to discuss the results of their examinations, the evaluations of our internal control over financial reporting, and the overall quality of our financial reporting.
Based on the reviews and discussions described above, we recommended to the Board of Directors, and the Board has approved that, the audited consolidated financial statements for the year ended December 31, 20092010 be included in Bristol-Myers Squibb’s Annual Report on Form 10-K for the year ended December 31, 20092010 for filing with the Securities and Exchange Commission.
In addition, we have confirmed there have been no new circumstances or developments since our respective appointments to the Committee that would impair any of our member’s ability to act independently.
The Audit Committee
Michael Grobstein, Chair
Lewis B. Campbell
Louis J. Freeh
Laurie H. Glimcher, M.D.
Leif Johansson
Alan J. Lacy
MANAGEMENT PROPOSALSITEM 3—ADVISORY VOTE ON THE COMPENSATION OF OUR NAMED EXECUTIVE OFFICERS
OurAs required by Section 14A of the Securities Exchange Act of 1934, as amended, we are providing stockholders the opportunity to advise the Compensation and Management Development Committee and the Board of Directors regarding the compensation of our named executive officers, as such compensation is described in its continuing reviewthe Compensation Discussion and Analysis (CD&A) section, the tabular disclosure regarding such compensation and the accompanying narrative disclosure, beginning on page 26.
Our executive compensation programs are designed to enable us to attract and retain talented employees capable of corporate governance matters,leading our business in the highly complex and after careful considerationcompetitive business environment in which we operate. We seek to accomplish this goal in a way that rewards performance and upon recommendationis aligned with our stockholders’ long-term interests. A significant part of the Committeeeach executive’s pay depends on Directorshis or her individual performance against financial and Corporate Governance, has concluded that itoperational objectives as well as key behavioral standards. In addition, a substantial portion of an executive’s compensation is in the best interestsform of equity awards that tie the executive’s compensation directly to creating stockholder value and achieving financial and operational results.
We strongly urge you to read the CD&A, which describes our compensation program in greater detail. Highlights include:
our pay-for-performance philosophy;
3-year and 5-year total shareholder return that exceeded our peer group average and the S&P 500 Index;
annual and long-term incentives that are 100% performance-based;
requiring a significant portion of an executives’ compensation to be at risk and tied to the creation of shareholder value;
having robust share ownership and share retention guidelines and prohibiting speculative and hedging transactions; and
appropriately mitigating risks through the use of various measure described in more detail on page 11.
We value input from our stockholders and have a strong record of engagement with our stockholders on compensation matters. As an advisory vote, this proposal is not binding on the company. The Compensation and Management Development Committee, however, will consider the outcome of the company’s stockholders to amend our Amended and Restated Certificate of Incorporation to (i) permit stockholders of 25% or more of our outstanding voting stock to call a special meeting of stockholders, (ii) removevote when making future executive compensation decisions.
Accordingly, the supermajority voting provision applicable to our common stockholders and (iii) remove the supermajority voting provisions applicable to our preferred stockholders. As each amendment to our Amended and Restated Certificate of Incorporation requires a different vote threshold to pass, we are asking our stockholders to vote on the following three management proposals separately. The Board of Directors unanimously recommends a vote “FOR” the approval of each of the amendments to our Amended and Restated Certificate of Incorporation (Items 3 through 5 to be presented at the 2010 Annual Meeting).
ITEM 3—APPROVAL OF AMENDMENT TO OUR AMENDED AND RESTATED CERTIFICATE OF INCORPORATION TO PERMIT STOCKHOLDERS TO CALL SPECIAL MEETINGS
Currently, our Amended and Restated Certificate of Incorporation provides that only the Chairman of the Board or a majority of the Board of Directors by written resolution may call a special meeting of stockholders. The ability of stockholders to call special stockholder meetings is increasingly considered an important aspect of good corporate governance. Last year, a stockholder proposal requesting that 10% of the holderscompensation of our outstanding stock be permitted to call special meetings received substantial support from our stockholders. After careful consideration, discussion with several investors and a review of our current stockholder base, the Board determined it was more appropriate to provide stockholders representing at least 25% in voting power of the outstanding stock of our company the right to call special meetings of stockholders. The Board believes that the 25% threshold is more appropriate for our company, because having the threshold as low as 10% would permit as little as two of our stockholders the right to call an unlimited number of special meetings. The Board believes that permitting such a small minority of stockholders the ability to call unlimited special meetings could be disruptive to our business and impose unnecessary costs on the company. Therefore, upon the recommendation of the Committee on Directors and Corporate Governance, the Board adopted resolutions (i) setting forth the proposed amendment to Article NINTH of our Amended and Restated Certificate of Incorporation to provide the holders of at least 25% in voting power of the outstanding shares of company stock the right to call special meetings and (ii) declaring the advisability of such amendment and recommending that our stockholders approve such amendment. The text of the second sentence of Article NINTH as proposed to be amended is set forth below with additions indicated by underlining and deletions by strike-out:
Except as otherwise required by law and subject to the rights under Article FOURTH hereof of the holders of any class or series of stock having a preference over the Common Stock as to dividends
or upon liquidation, special meetings of stockholders of the corporation may be called only by(i) the Chairman of the Boardor by the board of directorspursuant to a resolution approved by,(ii) a majority of the entire board of directors, or (iii) the Secretary of the corporation upon a written request of record holders of at least 25% in voting power of the outstanding shares of stock of the corporation made in accordance with, and subject to, all applicable provisions of the by-laws.named executive officers.
Exhibit B of this proxy statement sets forth the proposed Certificate of Amendment to the Amended and Restated Certificate of Incorporation. If this proposal is approved by the holders of a majority vote of our outstanding shares entitled to vote on the matter, the Certificate of Amendment will be filed with the Secretary of State of the State of Delaware. The Board of Directors has also adopted corresponding amendments to our Bylaws that describe the specific procedural requirements that must be followed by stockholders who want to call a special meeting. The Bylaw amendments will become effective only upon stockholder approval of the proposed amendment.
ITEM 4—APPROVAL OF AMENDMENT TO OUR AMENDED AND RESTATED CERTIFICATE OF INCORPORATION TO REMOVE THE SUPERMAJORITY VOTING PROVISION APPLICABLE TO COMMON STOCKHOLDERS
Our Board believes that generally, our governing documents should not contain supermajority voting provisions. At the 2005 Annual Meeting, our stockholders approved the Board’s recommendation to remove all of the supermajority voting provisions applicable to our common stockholders, except for one provision. The remaining supermajority voting provision is considered a stockholder friendly provision as it requires at least 75% of the holders of our outstanding stock to eliminate the annual election of directors and approve a classified Board structure. At the 2005 Annual Meeting, the Board believed it was appropriate to keep this supermajority voting provision as the Board was still in the stages of declassifying our Board following stockholder approval of the annual elections of directors. Last year, a stockholder proposal requesting the elimination of all supermajority voting provisions in our Amended and Restated Certificate of Incorporation received significant support. The Board, in its continuing review of corporate governance best practices and after careful consideration, determined that it is now appropriate to remove the remaining supermajority voting requirement as many investors view supermajority voting provisions as contrary to the principles of good corporate governance.
While the Board continues to believe that it is best to elect directors on an annual basis, the Board also believes that the annual election of directors has become a well-established best practice. The Board, therefore, does not believe removing this supermajority voting provision will result in our company returning to a classified board structure. Therefore, upon the recommendation of the Committee on Directors and Corporate Governance, the Board adopted resolutions (i) setting forth the proposed amendment to section (a) of Article EIGHTH of our Amended and Restated Certificate of Incorporation to remove the supermajority voting provision and (ii) declaring the advisability of such amendment and recommending that our stockholders approve such amendment. The text of the last sentence of section (a) of Article EIGHTH as proposed to be amended is set forth below with deletions indicated by strike-out:
Notwithstanding anything contained in this Certificate of Incorporation to the contrary, the affirmative vote of the holders of at least 75% of the outstanding shares of stock of the corporation entitled to vote generally in the election of directors, voting together as a single class, shall be required to alter, amend, adopt any provision inconsistent with or repeal this Article EIGHTH (a).
If our stockholders approve the deletion of this text, all supermajority voting provisions applicable to common stockholders will be eliminated. Exhibit C of this proxy statement sets forth the proposed Certificate of Amendment to the Amended and Restated Certificate of Incorporation. If this proposal is approved by the holders of at least 75% of the outstanding shares of our stock, the Certificate of Amendment will be filed with the Secretary of State of the State of Delaware.
ITEM 5—APPROVAL4—ADVISORY VOTE ON THE FREQUENCY OF AMENDMENT TO OUR AMENDED AND RESTATED CERTIFICATE OF INCORPORATION TO REMOVE THE SUPERMAJORITY VOTING PROVISIONS APPLICABLE TO PREFERRED STOCKHOLDERSADVISORY VOTE ON
Article FOURTHTHE COMPENSATION OF OUR NAMED EXECUTIVE OFFICERS
As required by Section 14A of the Securities Exchange Act of 1934, as amended, we are providing stockholders the opportunity to advise the Board of Directors regarding how frequently to conduct the advisory vote on executive compensation. Stockholders may indicate their preference for an annual, biennial (every two years) or triennial (every three years) advisory vote. We are required to hold at least once every six years an advisory vote to determine the frequency of the advisory stockholder vote on executive compensation.
There have been diverging views expressed on this question and the Board believes there is a reasonable basis for each of the options. Some believe that an annual vote is needed to give stockholders the opportunity to react promptly to emerging trends in compensation and to provide feedback before those trends become pronounced over time. In addition, an annual vote would provide stockholders the opportunity to evaluate, and advise on, individual compensation decisions made each year.
Others believe that a less frequent vote would be more beneficial because it would allow stockholders to focus on overall design issues rather than details of individual decisions and would better align with the goal of compensation programs to reward performance that promotes long-term shareholder value. In addition, several of our Amended and Restated Certificatelarge investors have advised us that a less frequent vote would lessen their burden of Incorporation provides forevaluating the compensation programs of a supermajority voting provision applicable solely tolarge number of companies.
After careful consideration, our preferred stockholders. As set forth above in Item 4, our Board of Directors has determined that itan advisory vote on executive compensation that occurs every three years (on a triennial basis) is the most appropriate alternative for BMS because such a vote would provide stockholders and advisory firms the opportunity to eliminate allevaluate our compensation philosophy and program on a more thorough, longer-term basis, consistent with our compensation philosophy. A three-year period would allow stockholders the opportunity to evaluate the effectiveness of our compensation program over the supermajority voting provisionstime frames that they are intended to generate performance and would also allow any changes to our compensation program to be in our Amendedplace long enough to evaluate whether the changes were effective. Additionally, a longer period between votes would provide the opportunity for stockholders to engage in more thoughtful analysis, would facilitate more meaningful dialogue between stockholders and Restated Certificate of Incorporation. Therefore, upon the recommendation of the Committee on Directors and Corporate Governance, the Board adopted resolutions (i) setting forthregarding the proposed amendmentCompany’s executive compensation practices and would allow the Board to more effectively implement changes to our Amended and Restated Certificate of Incorporation and the Certificate of the Designation, Preferences and Relative, Participating, Optional or Other Special Rights of the $2.00 Convertible Preferred Stock of the Corporation, which is attached as Appendix A to our Amended and Restated Certificate of Incorporation, to remove the supermajority voting provisions and (ii) declaring the advisability of such amendment and recommending that our stockholders approve such amendments.
The text of the first paragraph immediately following the heading “Preferred Stock” in Article FOURTH as proposed to be amended is set forth below with additions indicated by underlining and deletions by strike-out:
The affirmative vote of the holders of at leasttwo-thirdsa majorityof the Preferred Stock at the time outstanding voting only as a class shall be required to make effective any amendment to the Certificate of Incorporation or by-laws of the corporation altering materially any existing provisions of the Preferred Stock, or authorizing a class of preferred stock ranking prior to the Preferred Stock as to dividends or assets, and the affirmative vote of the holders of at least a majority of the Preferred Stock at the time outstanding voting only as a class shall be required to make effective any amendment to the Certificate of Incorporation of the corporation authorizing the issuance of or any increase in the authorized amount of any class of preferred stock ranking on a parity with or increasing the number of authorized shares of the Preferred Stock.compensation programs.
In addition, the text of the second paragraph of Section (c) of the Certificate of Designation as proposed to be amended is set forth below with additions indicated by underlining and deletions by strike-out:
So long as any shares of such series are outstanding, the consent of the holders of at leasttwo-thirdsa majority of the shares of such series at the time outstanding, given in person or by proxy, either in writing or at a meeting at which the holders of the shares of such series shallAs an advisory vote, separately as a class, shall be necessary for effecting the amendment, alteration or repeal of any provision of the Certificate of Incorporation of the Corporation, any certificate amendatory thereof or supplemental thereto, or the by-laws of the Corporation so as to affect materially any of the powers, preferences and rights of the shares of such series.
If our stockholders approve this proposal, all supermajority voting provisions applicable to preferred stockholders will be eliminated. Exhibit D of this proxy statement sets forth the proposed Certificate of Amendment to the Amended and Restated Certificate of Incorporation. If this proposal is approved by bothnot binding on the holderscompany. However, our Board will take into account our stockholders’ preferences when considering the frequency of a majorityfuture advisory votes on executive compensation.
Accordingly, the Board of our outstanding shares and at least two-thirds of our outstanding shares of preferred stock,Directors unanimously recommends that the Certificate of Amendment willadvisory vote on executive compensation be filed with the Secretary of State of the State of Delaware.conducted every THREE years.
STOCKHOLDER PROPOSALS
We expect the following stockholder proposals (Items 65 through 8)7) to be presented at the 20102011 Annual Meeting. The Board of Directors has recommended a vote against these proposals for the policy reasons as set forth following the proposal. The stock holdings of the proponent will be provided upon request to the Secretary of Bristol-Myers Squibb.
ITEM 6—5—STOCKHOLDER PROPOSAL ON EXECUTIVE COMPENSATION DISCLOSURE
The proponent of this resolution is Evelyn Y. Davis of 2600 Virginia Avenue N.W., Suite 215, Washington, DC 20037.
RESOLVED: “That the shareholders recommend that the Board take the necessary steps that Bristol-Myers Squibb specifically identify by name and corporate title in all future proxy statements those executive officers, not otherwise so identified, who are contractually entitled to receive in excess of $500,000 annually as a base salary, together with whatever other additional compensation bonuses and other cash payments were due them.”
REASONS: “In support of such proposed Resolution it is clear that the shareholders have a right to comprehensively evaluate the management in the manner in which the Corporation is being operated and its resources utilized.” “At present only a few of the most senior executive officers are so identified, and not the many other senior executive officers who should contribute to the ultimate success of the Corporation.” “Through such additional identification the shareholders will then be provided an opportunity to better evaluate the soundness and efficacy of the overall management.”
“Last year the owners of 168,590,71799,084,372 shares, representing approximately 11.8%8.3% of shares voting, voted FOR this proposal.”
“If you AGREE, please mark your proxy FOR this proposal.”
Board of Directors’ Position
The Board of Directors recommends a vote “AGAINST” this proposal for the following reasons:
The Board of Directors believes that this proposal is unnecessary. The Securities and Exchange Commission (SEC) recentlyhas revised and expanded executive compensation disclosure requirements, adopting a principles-based approach. In compliance with the SEC requirements, the Compensation Disclosure and Analysis section beginning on page 25,26, of this Proxy Statement, details the company’s philosophy and objectives in determining executive compensation and the various compensation methods and analyses used to accomplish those objectives. This Proxy Statement also discloses in great detail the compensation of the company’s most highly compensated employees as required by the SEC rules. The Summary Compensation Table on page 46,47, for example, sets forth their salary, bonus, stock and option awards, non-equity incentive plan compensation, change in pension value and all other compensation. Therefore, the Board believes that the executive compensation disclosures provided in this Proxy Statement provide stockholders with detailed information to effectively evaluate executive compensation at our company.
The Board believes that providing the additional information requested by the proponentProponent would place our company at a competitive disadvantage in that it would require us to disclose detailed compensation information about a larger group of employees that is not otherwise available. The Board believes that requiring our company to make disclosures other companies are not required to make could be detrimental to the company by deterring talented individuals from joining our company.
Furthermore, the Board believes that the proposal would impose unwarranted costs and administrative burdens on the company with little or no associated benefit to stockholders and, therefore, is not in the best interest of the company or its stockholders.
Accordingly, the Board of Directors unanimously recommends a vote “AGAINST” this proposal.
ITEM 7—6—STOCKHOLDER PROPOSAL ON SHAREHOLDER ACTION BY WRITTEN CONSENT
The proponent of this resolution is Kenneth Steiner of 14 Stoner Ave., 2M, Great Neck, New York 11021.
RESOLVED, Shareholders hereby request that our board of directors undertake such steps as may be necessary to permit shareholders to act by the written consent by shareholders entitled to cast the minimum number of votes that would be necessary to authorize the action at a majoritymeeting at which all shareholders entitled to vote thereon were present and voting (to the fullest extent permitted by law).
We gave greater than 49%-support to a 2010 proposal on this same topic. Hundreds of our shares outstanding in compliance with applicable laws.major companies enable shareholder action by written consent.
Taking action by written consent in lieu of a meeting is a mechanismmeans shareholders can use to raise important matters outside the normal annual meeting cycle.
Limitations on shareholders’ rights to act by written consent are considered takeover defenses because they may impede the ability of a bidder to succeed in completing a profitable transaction for us or in obtaining control of the board that could result in a higher price for our stock. Although it is not necessarily anticipated that a bidder will materialize, that very possibility presents a powerful incentive for improved management of our company.
A study by Harvard professor Paul Gompers supports the concept that shareholder dis-empowering governance features, including restrictions on shareholder ability to act by written consent, are significantly correlatedrelated to a reduction inreduced shareholder value.
The meritsmerit of this Shareholder Action by Written Consent proposal should also be considered in the context of the need for improvementsimprovement in our company’s 20092010 reported corporate governance status:
We had no shareholder right call a special meeting,The Corporate Librarywww.thecorporatelibrary.com, an independent board chairman, cumulative voting or vote oninvestment research firm rated our company “D” with “High Governance Risk,” and “Very High Concern” in executive pay. A 75% votepay—$18 million for James Cornelius and $10 million for Elliot Sigal. Mr. Cornelius realized more than $8 million from the vesting of stock in 2009 and was requiredentitled to removemore than $30 million if he were terminated following a director for cause. Approvalchange of 75% of shares was required to amend Article Eighth (Directors)control. Executive pay practices were not aligned with shareholder interest.
Togo West, one of our charter.newest directors, was marked a “Flagged (Problem) Director” by The Corporate Library due to his Krispy Kreme and AbitibiBowater directorships prior to both bankruptcies. Yet Mr. West and Louis Freeh (our highest negative vote-getter) were on our key Executive Pay and Nomination Committees. Three directors with long-tenure (Laurie Glimcher, Leif Johansson and Lewis Campbell) were assigned to 7 of 17 seats on our key board committees—independence concern.
The above concerns shows there is need for improvement. Please encourage our board to respond positively to this proposal to enable shareholder action by written consent—Yes on 7.6.
Board of Directors’ Position
The Board of Directors recommends a vote “AGAINST” this proposal for the following reasons:
The Board believes that this proposal is not in the best interests of the stockholders because, unlike meetings of stockholders, action by written consent would result in certain stockholders being denied the ability to vote or otherwise have a say on proposed stockholder actions. Action by written consent would enable a majority of our stockholders to take action on a proposal without the benefit of hearing the views, questions and arguments of other stockholders. In addition, action by written consent eliminates the need for notice to be given to stockholders about a proposed action, and therefore, certain stockholders may not be informed about the proposed action until after the action has already been taken. This would deny these stockholders the ability to determine whether to exercise their rights, such as by expressing their views, encouraging the Board to reconsider the matter and voting on the proposed action. The Board, therefore, believes that this proposal could adversely affect the conduct of stockholder business by resulting in our taking of an action that we otherwise would not have taken if our stockholders were afforded the opportunity to discuss, debate and vote on the matter.
The Board also believes that adoption of this proposal is unnecessary because the company is committed to high standards of corporate governance and has already taken a number of steps to achieve greater transparency and accountability to stockholders. These corporate governance practices and policies cover a wide range of matters and are described beginning on page 5 of this Proxy Statement under the heading “Corporate Governance and Board Matters.” Our Board continually reassesses our corporate governance practices to identify additional steps to further benefit our stockholders. In this regard, our Board has agreed to adopt an advisory vote on executive
compensation (“say on pay”)recommended, and our stockholders approved, at the 20112010 Annual Meeting of Stockholders. In addition, our corporate governance practices will be further strengthened if our stockholders approve management’s proposalsStockholders, amendments to amend our Amended and Restated Certificate of Incorporation to eliminate all supermajority provisions applicable to common stockholders and to permit stockholders holding a minimum of 25% of our outstanding shares of common stock to call a special meeting of stockholders. The Board believes that holding meetings whereby all stockholders may discuss the proposed actions and vote their shares is the best way for stockholders to take action. Not only do stockholder meetings provide stockholders with a much more meaningful way to participate in proposed actions, but it permits a more rigorous and careful consideration of proposed actions by both the Board and the stockholders. Adoption of this proposal, however, would mean that certain stockholders would not be able to debate the issues nor will they be able to vote on the proposed action. The Board, therefore, believes that action by written consent would undermine stockholder democracy and is not in the best interests of the stockholders.
Accordingly, the Board of Directors unanimously recommends a vote “AGAINST” the proposal.
ITEM 8—7—STOCKHOLDER PROPOSAL ON REPORT ON ANIMAL USEPHARMACEUTICAL PRICE RESTRAINT
The proponents of this resolution are Ned EisenbergTrinity Health, 27870 Cabot Drive, Novi, Michigan 48377 as lead-filer and 13 other religious investors who are members of P.O. Box 230091, New York, NY 10023The Interfaith Center for Corporate Responsibility.
WHEREAS:
The cost of brand name drugs, some of them our Company’s, have skyrocketed in this country in recent years;
The Government’s General Accountability Office (GAO) found that between 2000-2008, 416 brand-name drugs had “extraordinary price increases”; most of these increases ranged from 100% to 499%;
Medco’s 2010 Drug Trend Report found that, while generic drug prices increased 0.3%, “inflation in branded drugs accelerated to an all-time high of 9.2%” and James Moranthat the prices of P.O. Box 15889, Seattle, WA 98115.specialty drugs increased 14.7%;
RESOLVED: shareholders encourage Bristol-Myers Squibb Company (“Bristol-Myers Squibb”)The Office of Actuary, Centers for Medicare and Medicaid Services projects that prescription drug expenditures will increase in 2018 92.7% from 2008 expenditures, exceeding all major categories of national health expenditures. It states: “Prescription drug spending is expected to increase its corporate social responsibility and transparency aroundbe the usefast growing component of animals in research and product testing,Medicare over the projection period”;
AARP’s Public Policy Institute reported that the price of brand name prescriptions most widely used by including information on animal use in its sustainability report (“Report”). We encourage the Report to include non-proprietary information, as follows: (1) species, numbers, and general purpose of each use (e.g., research and development, efficacy testing, or toxicity testing), and (2) Bristol-Myers Squibb’s effortsMedicare beneficiaries increased by 9.7% in the preceding year12 months ending with March 2010 and futurewas much higher than the rate of increase observed during any of the prior eight years (2002-2009). While inflation rose 0.3% during his period, price increases for such drugs ranged from 5.3-9.3%;
AARP has also stated that the positive goals towards reducing and replacing animal use.of the new health care reform law “could be eroded over the years if escalating drug prices are not addressed”;
SUPPORTING STATEMENT
Companies using animals for product development and testing have an ethical imperative to address animal use, since 43%While passage of Americans oppose the use of animals for research.1 Responding to societal concerns, several pharmaceutical companies now disclose information, including development and implementation of methods that replace, reduce, or refine animal use. To address public and shareholder concerns, Bristol-Myers Squibb can make this information available in the Report.
The Report would be ideal for providing animal use information because it outlines Bristol-Myers Squibb’s social priorities and progress from environmental impacts to philanthropy and community service projects. This same level of commitment and transparency demonstrated for those areas can be extended to animal use.
In addition to the ethical imperative,health reform legislation was a major achievement, there are scientificongoing concerns as to its long-term affordability and financial imperativesaccountability for moving away from animal use. Astonishingly, 92%controlling costs. Failure to control costs could undermine the goals of drugs deemed safehealth care reform, i.e. accessible and effectiveaffordable health care for all;
This resolution’s sponsors are not satisfied that the Company has made a clear case offering fiscal and moral justification for such exorbitant price increases. Neither has it given sufficient assurances that the present pattern of increases that far exceed the Consumer Price Index will not continue.
RESOLVED: Shareholders request the Board of Directors create and implement a policy of price restraint on branded pharmaceuticals, utilizing a combination of approaches to keep drug prices at reasonable levels, such as an increase that would not exceed the previous year’s Consumer Price Index, and report to shareholders by September 2011 on changes in animals, fail when tested in humans.2 In the 8% of FDA-approved drugs, half are later relabeled or withdrawn due to unanticipated, severe, adverse effects. A 96% failure rate not only challenges the reliability of animal experiments to predict human safetypolicies and efficacy, it creates enormous risks of litigation, adverse publicity,pricing procedures for pharmaceutical products (withholding any competitive information, and wasted resources. Primary reasons for this significant failure rate are the anatomical and physiological differences between humans and other species. To deliver safer, more effective products, pharmaceutical companies need to focus on experimental models with greater human relevance. As highlighted by a 2007 National Academy of Sciences report,at reasonable cost).3 advances in many areas of science-
|
toxicogenomics, bioinformatics, systems biology, epigenetics, and computational toxicology- are making it possible to replace animal toxicity tests with non-animal methods. These human-based methods confer numerous advantages including quicker and more economical product development and approval, reduced incidence of adverse effects, improved efficacy, and reduced animal use and suffering.
Given the ethical and scientific implications of animal use for drug development and testing, we urge shareholders to vote in favor of this proposal for Bristol-Myers Squibb’s consideration to increase transparency about its animal use and replacement efforts in the Report.
Board of Directors’ Position
The Board of Directors recommends a vote “AGAINST” this proposal for the following reasons:
TheOur Board of Directors firmly believes that this proposalprescription drugs are so important that everyone should have access to them. We believe the best approach to ensure broad access to affordable medicines for the uninsured and underinsured is unnecessary becausethrough expanded coverage, not price restraints. Accordingly, we already include informationrecommend a vote AGAINST the proposal.
We have been, and remain, committed to facilitating access to our medicines to further our mission to help patients prevail over serious diseases. We are also sensitive to concerns about rising costs of health care, including pharmaceuticals, and access to medicines. Consequently, we price our medicines based on animalthe value they deliver to individuals and society. We also recognize that there are significant unmet medical needs that require the development of additional innovative treatments. Therefore, our goal is to ensure access to currently approved medicines while fueling the development of medicines for the future.
New drug development is a difficult and risky effort. Only 1 of every 5,000 -10,000 potential medicines investigated by U.S. research-based pharmaceutical companies makes it through the R&D process and is approved for patient use by the U.S. FDA.1 And only 2 in our sustainability report, including our commitment10 approved medicines bring in enough revenue to reducerecoup the average cost of development.2 Thus the very few drug candidates that receive FDA approval and replace animals, where feasible. Therefore,become commercially successful must support the Board believes that publishing an additional report to stockholders would not be a good use of company resources.entire R&D enterprise.
We believesupport expanded access to insurance coverage that as a research-based biopharmaceutical company seekingwould give covered patients access to discover, developdiscounts on medicines their coverage plan negotiates from manufacturers and deliver innovative medicines to patients,pharmacies. In addition, we have both legal and ethical obligations to ensurenote that the safety and efficacy of our medicines. At times, to ensure fulfillment of these obligations, research or testing methodsnew health care reform law that includewas passed last year includes provisions that will expand health care coverage, including prescription drug coverage, for the use of animals is required. All biopharmaceutical and healthcare companies are required by law to demonstrate the safety and efficacy of their products in animals. Testing with animals, where required,uninsured.
We will continue to work with our various constituencies, including our stockholders, to ensure broad access to affordable medicines. Individuals who cannot afford our medicines and have no other means of coverage, public or private, may be a componenteligible to be provided with our medicines, at no charge, through the Bristol-Myers Squibb Patient Assistance Program Foundation, Inc. and other company sponsored patient assistance programs. We estimate that in 2010 alone, we provided more than $458 million worth of medicines to more than 263,000 patients at no cost to these patients. In addition, the company makes contributions to independent co-pay programs to help patients afford their medicines.
The Board strongly believes that we should not be compelled to adopt any formal policy regarding pricing increases. In this fast-paced and highly competitive industry, we need the flexibility to price our products appropriately so that we may invest aggressively in the research and development efforts.
Our sustainability report, availableof promising new and innovative medicines. This investment will continue to increase as we accelerate the growth of our pipeline of new cures and therapies in areas of significant unmet medical need. This is fundamental to our continued success and to the patients who rely on our website at www.bms.com/sustainability, details our long-standing, highly regarded animal testing program that is committed to reducing reliance on animal testing methods, promotes the development, validation and use of non-animal tests, and provides the highest level of care when use of animals is required.
As stated in our sustainability report, it is our policy to insist that our scientists always consider replacing the use of animals by other methods, reducing the number of animals used and refining procedures to enhance animal welfare (also known as the 3Rs—replace, reduce and refine). We are committed to developing non-animal tests, and using them whenever possible. We have supported development of alternatives to animal research both internally, as part of the discovery and development process, and externally with over one million dollars of funding to other institutions. Employees involved in any aspect of our animal research program attend regular training that addresses humane care, use and respect for all research animals.
In addition, all of our procedures that involve the use and care of animals must comply with applicable governmental regulations and our internal corporate animal care and use directives. We continue to meet or exceed the high standards of care and humaneness set by the National Research Council of the National Academy of Sciences, the National Institutes of Health and the U.S. Department of Agriculture as mandated under the Animal Welfare Act. All of our facilities and programs involved with the care and use of animals are periodically subjected to rigorous inspection by company auditors and government inspectors to ensure compliance with applicable standards.
Our animal testing program integrates our firm commitment to reduce use of animals, to develop, where feasible, alternatives to animal use and to pursue humane care of the animals we use. The Board believes that the disclosures already contained in sustainability report provide our stockholders with sufficient information to assess our animal testing program. Therefore, the Board believes that publishing an additional report to stockholders beyond what is already published in our sustainability report is unnecessary and would not be in the best interests of our stockholders.medicines.
Accordingly, the Board of Directors unanimously recommends a vote “AGAINST” the proposal.
| 1 | Based on data from Tufts University, Tufts Center for the Study of Drug Development (1995). |
| 2 | John E. Calfee, “White Paper on Pharmaceutical Market Competition Issues,” American Enterprise Institute, June 2, 2008. |
ADVANCE NOTICE PROCEDURESVOTING SECURITIES AND PRINCIPAL HOLDERS
At the close of business on March 10, 2011, there were 1,704,681,382 shares of $0.10 par value common stock and 5,268 shares of $2.00 convertible preferred stock outstanding and entitled to vote.
Common Stock Ownership by Directors and Executive Officers
The following table sets forth, as of February 15, 2011, beneficial ownership of shares of our common stock by each director, each of the named executive officers and all directors and executive officers as a group. Shares are beneficially owned when an individual has voting and/or investment power over the shares or could obtain voting and/or investment power over the shares within 60 days. Voting power includes the power to direct the voting of the shares and investment power includes the power to direct the disposition of the shares. Unless otherwise noted, shares listed below are owned directly or indirectly with sole voting and investment power. None of our directors and executive officers, individually or as a group, beneficially owns greater than 1% of our outstanding shares of common or preferred stock.
Name | Bristol-Myers Squibb Company | |||||||||||
| Total Common Shares Owned(1) | Common Shares Underlying Options or Stock Units(2) | Common Shares Underlying Deferred Share Units(3) | ||||||||||
L. Andreotti | 2,092,884 | 1,896,795 | 0 | |||||||||
C. A. Bancroft | 248,192 | 221,728 | 0 | |||||||||
L. B. Campbell | 41,087 | 18,181 | 20,796 | |||||||||
J. M. Cornelius | 3,520,174 | 3,137,770 | 13,567 | |||||||||
L. J. Freeh | 31,779 | 2,500 | 29,279 | |||||||||
L. H. Glimcher, M.D. | 77,396 | 18,181 | 59,215 | |||||||||
M. Grobstein | 33,961 | 0 | 30,578 | |||||||||
A. C. Hooper | 1,029,015 | 915,266 | 0 | |||||||||
L. Johansson | 61,396 | 18,181 | 41,215 | |||||||||
A. J. Lacy | 27,732 | 0 | 25,427 | |||||||||
S. Leung | 606,362 | 562,354 | 0 | |||||||||
V. L. Sato, Ph.D. | 27,291 | 0 | 27,291 | |||||||||
E. Sigal, M.D., Ph.D. | 1,533,997 | 1,375,255 | 22,757 | |||||||||
T. D. West, Jr. | 22,389 | 0 | 22,389 | |||||||||
R. S. Williams, M.D. | 36,843 | 0 | 36,343 | |||||||||
All Directors and Executive Officers as a Group(4) | 12,088,038 | 10,432,143 | 328,854 | |||||||||
| (1) | Consists of direct and indirect ownership of shares, including vested and unvested restricted stock, shares credited to the accounts of the executive officers under the Bristol-Myers Squibb Company Savings and Investment Program, stock options that are currently exercisable or exercisable within 60 days, restricted stock units that vest within 60 days, the target number of market share units that will vest within 60 days and deferred share units. |
| (2) | Consists of shares underlying stock options that are currently exercisable, stock options that will be exercisable within 60 days, restricted stock units that will vest within 60 days and the target number of market share units that will vest within 60 days. None of these shares have any voting rights. |
| (3) | Consists of deferred share units that are valued according to the market value and shareholder return on equivalent shares of common stock. Deferred share units have no voting rights. |
| (4) | Includes 22 individuals. |
Principal Holders of Voting Securities
The following table sets forth information regarding beneficial owners of more than 5 percent of the outstanding shares of our common stock. There are no beneficial owners of more than 5 percent of the outstanding shares of our preferred stock.
Name | Number of Shares Beneficially Owned | Percent of Class | ||||||
BlackRock, Inc. 40 East 52nd Street New York, NY 10022 | 133,819,873 | (1) | 7.82 | %(1) | ||||
Capital World Investors 333 South Hope Street Los Angeles, CA 90071 | 131,770,500 | (2) | 7.7 | %(2) | ||||
| (1) | This information is based on the Schedule 13G/A dated February 3, 2011 filed by BlackRock, Inc. with the Securities and Exchange Commission reporting beneficial ownership as of December 31, 2010. The reporting person has sole voting power with respect to all 133,819,873 shares. |
| (2) | This information is based on the Schedule 13G dated February 14, 2011 filed by Capital World Investors with the Securities and Exchange Commission reporting beneficial ownership as of December 31, 2010. The reporting person has sole voting power with respect to 106,970,500 shares and sole dispositive power with respect to all 135,563,389 shares. |
Section 16(a) Beneficial Ownership Reporting Compliance
Under our Bylaws, no business, including the nomination of directors, may be brought before an annual meeting except as set forth in the noticeSection 16(a) of the meeting or as otherwise brought beforeSecurities Exchange Act of 1934, our directors, executive officers and the meeting by or at the directionbeneficial holders of the Board or by a stockholder entitled to vote who has delivered notice to our company containing certain information set forth in our Bylaws, not less than 90 but not more than 120 days from10% of our common stock are required to file reports of ownership and changes in ownership with the anniversaryU.S. Securities and Exchange Commission. To the best of the prior year’s Annual Meeting. For our knowledge, during 2010 Annual Meeting, we must receive this notice between Januaryall applicable Section 16(a) filing requirements were met, except that, due to administrative errors, Forms 4 2011 and February 3, 2011. These requirements are separate and distinct from and in additionwere filed late for Joseph Caldarella relating to the SEC requirements that a stockholder must meet to have a stockholder proposal included in our proxy statement.
Our Bylaws are availablevesting of 2,378 restricted stock units on our website at www.bms.com/ourcompany/governance. In addition, a copy of the Bylaw provisions discussed above may be obtained by writing to us at our principal executive offices, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Secretary.
Stockholder proposalsMarch 3, 2010 and for Beatrice Cazala relating to our 2011 Annual Meetingthe vesting of Stockholders must be received by us at our principal executive offices, Bristol-Myers Squibb Company, 345 Park Avenue, New York, New York 10154, attention: Secretary, no later than November 22,2,681 restricted stock units on March 12, 2010. Stockholders are encouraged to contact the Office of the Corporate Secretary prior to submitting a stockholder proposal or any time they have a concern. At the direction of the Board of Directors, the Office of the Corporate Secretary acts as corporate governance liaison to stockholders.
Categorical Standards of Independence
The Board has adopted the following categorical standards for determining which relationships will be considered immaterial:
| a) | an immediate family member of the director is or has been employed by the Company, provided that such family member is not, and has not been for at least a period of three years, an executive officer of the Company; |
| b) | more than three years has elapsed since: i) the director was employed by the Company; ii) an immediate family member of the director was employed by the Company as an executive officer; or iii) an executive officer of the Company was on the board of directors of a company that employed either the director or an immediate family member of the director as an executive officer; |
| c) | the director, or an immediate family member of the director, received $120,000 or less in any year in direct compensation from the Company (other than director’s fees or compensation that was deferred for prior service with the Company); |
| d) | more than three years has elapsed since i) the director has been a partner with or employed by the Company’s independent auditor or ii) an immediate family member personally worked on the Company’s audit as a partner or employee of the Company’s independent auditor; |
| e) | the director has an immediate family member who i) is an employee of, but not a partner of, the independent auditor and ii) does not personally work on the Company’s audit; |
| f) | the director of the Company, or an immediate family member of a director, is a director, an executive officer or an employee of another company that makes payment to, or receives payment from, the Company for property or services in an amount which, in any single fiscal year, does not exceed the greater of $1 million or 2% of such other company’s consolidated gross revenues; |
| g) | the director of the Company and/or an immediate family member of the director directly or indirectly owns, in the aggregate, 10% equity interest or less in another company that makes payment to, or receives payment from, the Company for property or services; and |
| h) | the director of the Company is a director or an executive officer of a charitable organization or non-profit organization, and the Company’s, or the Bristol-Myers Squibb Foundation’s discretionary charitable contributions to the organization, in aggregate, in any single fiscal year within the preceding three years, do not exceed the greater of $1 million or 2% of that organization’s consolidated gross revenues. |
CERTIFICATE OF AMENDMENT
TO THE
AMENDED AND RESTATED CERTIFICATE OF INCORPORATION
OF
BRISTOL-MYERS SQUIBB COMPANY
Pursuant to Section 242
of the General Corporation Law of the State of Delaware
Bristol-Myers Squibb Company, a corporation duly organized and existing under the General Corporation Law of the State of Delaware (the “Corporation”), does hereby certify that:
1. The Amended and Restated Certificate of Incorporation of the Corporation (the “Certificate of Incorporation”) is hereby amended by deleting the second sentence of Article NINTH of the Certificate of Incorporation in its entirety and inserting the following in lieu thereof:
Except as otherwise required by law and subject to the rights under Article FOURTH hereof of the holders of any class or series of stock having a preference over the Common Stock as to dividends or upon liquidation, special meetings of stockholders of the corporation may be called only by (i) the Chairman of the board of directors, (ii) a majority of the entire board of directors, or (iii) the Secretary of the corporation upon a written request of record holders of at least 25% in voting power of the outstanding shares of stock of the corporation made in accordance with, and subject to, all applicable provisions of the Bylaws.
The foregoing amendment was duly adopted in accordance with the provisions of Section 242 of the General Corporation Law of the State of Delaware.
IN WITNESS WHEREOF, the Corporation has caused this Certificate of Amendment to be executed by a duly authorized officer on this day of , 20 .
| ||
| ||
| ||
CERTIFICATE OF AMENDMENT
TO THE
AMENDED AND RESTATED CERTIFICATE OF INCORPORATION
OF
BRISTOL-MYERS SQUIBB COMPANY
Pursuant to Section 242
of the General Corporation Law of the State of Delaware
Bristol-Myers Squibb Company, a corporation duly organized and existing under the General Corporation Law of the State of Delaware (the “Corporation”), does hereby certify that:
1. The Amended and Restated Certificate of Incorporation of the Corporation (the “Certificate of Incorporation”) is hereby amended by deleting in its entirety the last paragraph in section (a) of Article EIGHTH of the Certificate of Incorporation.
The foregoing amendment was duly adopted in accordance with the provisions of Section 242 of the General Corporation Law of the State of Delaware.
IN WITNESS WHEREOF, the Corporation has caused this Certificate of Amendment to be executed by a duly authorized officer on this day of , 20 .
| ||
| ||
| ||
CERTIFICATE OF AMENDMENT
TO THE
AMENDED AND RESTATED CERTIFICATE OF INCORPORATION
OF
BRISTOL-MYERS SQUIBB COMPANY
Pursuant to Section 242
of the General Corporation Law of the State of Delaware
Bristol-Myers Squibb Company, a corporation duly organized and existing under the General Corporation Law of the State of Delaware (the “Corporation”), does hereby certify that:
1. The Amended and Restated Certificate of Incorporation of the Corporation (the “Certificate of Incorporation”) is hereby amended by deleting “two-thirds” from the first paragraph immediately following the heading “Preferred Stock” in Article FOURTH of the Certificate of Incorporation and inserting “a majority” in lieu thereof.
2. The Certificate of the Designation, Preferences and Relative, Participating, Optional or Other Special Rights of the $2.00 Convertible Preferred Stock of the Corporation (the “Certificate of Designation”), which is attached as Appendix A to the Certificate of Incorporation of the Corporation, is hereby amended by deleting “two-thirds” from the second paragraph of Section (c) of the Certificate of Designation and inserting “a majority” in lieu thereof.
The foregoing amendments were duly adopted in accordance with the provisions of Section 242 of the General Corporation Law of the State of Delaware.
IN WITNESS WHEREOF, the Corporation has caused this Certificate of Amendment to be executed by a duly authorized officer on this day of , 20 .
| ||
| ||
| ||
DIRECTIONS TO OUR PLAINSBORO OFFICE AT
777 SCUDDERS MILL ROAD
PLAINSBORO, NJ 08536
By Car:
From New York:
Take the Lincoln Tunnel.
Take the New Jersey Turnpike South/Newark ExitExit.
Exit Left onto I-95 South.
Merge onto NJ-18 North Via Exit 9 toward US-1/New Brunswick/Princeton.
Merge onto US-1 South toward Trenton.
Take ramp onto Scudders Mill Rd.
Our offices are approximately 1 1/2 mile on the left side of the road.
From Philadelphia:
Take I-95 North.
Merge onto US-1 North via Exit 67A toward New Brunswick.
Turn Slight Right onto Scudders Mill Road.
Our offices are approximately 1 1/2 mile on the left side of the road.
By Train:
New Jersey Transit and Amtrak train service is available to Princeton Junction, New Jersey. Our Plainsboro office is approximately a 10 minute car drive from the station.
Parking:
Free parking for stockholders attending the 20102011 Annual Meeting is available. Please go directly to the parking area reserved for stockholders.
Y O U R V O T E I S I M P O R T A N T
P L E A S E V O T E Y O U R P R O XY


P.O. BOX 4000 PRINCETON, NJ 08540 | VOTE BY INTERNET | |
Use the internet to transmit your voting instructions and for electronic delivery of information up until 11:59 P.M. Eastern Time, either on (i) Thursday, April | ||
ELECTRONIC DELIVERY OF FUTURE STOCKHOLDER COMMUNICATIONS If you would like to reduce the costs incurred by | ||
| VOTE BY PHONE | ||
| Use any touch-tone telephone to transmit your voting instructions up until 11:59 P.M. Eastern Time, either on (i) Thursday, April | ||
| VOTE BY MAIL | ||
| Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Bristol-Myers Squibb Company, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717. We recommend you mail your proxy by April | ||
| TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: | ||||
| M31837-P07625-Z54874 | KEEP THIS PORTION |
| THIS PROXY CARD IS VALID ONLY WHEN SIGNED AND DATED. | DETACH AND RETURN THIS PORTION ONLY | |||||||
BRISTOL-MYERS SQUIBB COMPANY | ||||||||||||||||||||||||||||||||
THE BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” | ||||||||||||||||||||||||||||||||
| EACH DIRECTOR UNDER ITEM 1. | ||||||||||||||||||||||||||||||||
| For | Against | Abstain | ||||||||||||||||||||||||||||||
| 1. | Election of Directors | |||||||||||||||||||||||||||||||
Nominees: | ||||||||||||||||||||||||||||||||
| 1A) | ||||||||||||||||||||||||||||||||
| ¨ | ¨ | ¨ | |||||||||||||||||||||||||||||
1B) | L. B. Campbell | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
1C) | J. M. Cornelius | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| L. J. Freeh | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| L. H. Glimcher, M.D. | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| 1F) | M. Grobstein | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| 1G) | L. Johansson | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| 1H) | A. J. Lacy | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| 1I) | V. L. Sato, Ph.D. | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| 1J) | E. Sigal, M.D., Ph.D. | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| 1K) | T. D. West, Jr. | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
| 1L) | R. S. Williams, M.D. | ¨ | ¨ | ¨ | ||||||||||||||||||||||||||||
|
|
|
|
| ||||||||||||||||||||||||||||
|
|
|
|
| ||||||||||||||||||||||||||||
|
|
|
|
| ||||||||||||||||||||||||||||
|
|
|
|
| ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||
For address changes and/or comments, please check this box and write them on the back where indicated. | ¨ | |||||||||||||||||||||||||||||||
| ¨ | ¨ | |||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
| |||||||||||||||||||||||||
| Yes | No | |||||||||||||||||||||||||||||||
| For | Against | Abstain | ||||||||||
| THE BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” ITEMS 2 AND 3. | ||||||||||||
| 2. | Ratification of the Appointment of Independent Registered Public Accounting Firm | ¨ | ¨ | ¨ | ||||||||
| 3. | Advisory Vote on | ¨ | ¨ | ¨ | ||||||||
| 3 Years | 2 Years | 1 Year | Abstain | |||||||||
| THE BOARD OF DIRECTORS RECOMMENDS A VOTE FOR “3 YEARS” ON ITEM 4. | ||||||||||||
| 4. | Advisory Vote on the Frequency of the Advisory Vote on the Compensation of our Named Executive Officers | ¨ | ¨ | ¨ | ¨ | |||||||
| For | Against | Abstain | ||||||||||
| THE BOARD OF DIRECTORS RECOMMENDS A VOTE “AGAINST” ITEMS 5, 6 AND 7. | ||||||||||||
| 5. | Executive Compensation Disclosure | ¨ | ¨ | ¨ | ||||||||
| 6. | Shareholder Action by Written Consent | ¨ | ¨ | ¨ | ||||||||
| 7. | Pharmaceutical Price Restraint | ¨ | ¨ | ¨ | ||||||||
| Note: Please sign as name appears on this card. Joint owners should each sign. When signing as attorney, executor, administrator, trustee or guardian, please give full title as such. |
| Signature [PLEASE SIGN WITHIN BOX] | Date | |||||||||||||||||||||||
| Signature | Date |
|
| |||||||||||||||||||||||
ADMISSION TICKET
20102011 Annual Meeting of Stockholders
Tuesday, May 4, 20103, 2011
10:00 A.M.
Bristol-Myers Squibb Company
777 Scudders Mill Road
Plainsboro, New Jersey
PHOTO IDENTIFICATION WILL BE REQUIRED
This is your admission ticket to the meeting.
This ticket admits only the stockholder(s) listed on the reverse side of this card and is not transferable.
Bristol-Myers Squibb Company is located at 777 Scudders Mill Road, Plainsboro, New Jersey.
Directions to the facility can be found on the inside back cover of the Proxy Statement or you can call the company at 609-897-2000.
Free parking for stockholders attending the 20102011 Annual Meeting is available at Bristol-Myers Squibb.
Important Notice Regarding Internetthe Availability of Proxy Materials for the 20102011 Annual Meeting:
The Notice of 20102011 Annual Meeting, Proxy Statement and Annual Report are available at www.proxyvote.com.
M20753-P91264-Z51999
| M31838-P07625-Z54874 |
THIS PROXY IS SOLICITED ON BEHALF OF THE BOARD OF DIRECTORS FOR THE ANNUAL MEETING OF STOCKHOLDERS, MAY
The undersigned hereby appoints
This proxy also provides voting instructions for shares held by the Trustee of the Bristol-Myers Squibb Company Savings and Investment Program, the Bristol-Myers Squibb Company Employee Incentive Thrift Plan, and the Bristol-Myers Squibb Puerto Rico, Inc. Savings and Investment Program,
When properly executed, your proxy will be voted as you indicate, or where no contrary indication is made, will be voted FOR Items1, 2 and 3, for 3 YEARS for Item 4, | ||||||||||||
Address Changes/Comments: |
| |||||||||||
| ||||||||||||
(If you noted any Address Changes/Comments above, please mark corresponding box on the reverse side.) | ||||||||||||